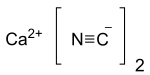Chemical compound
Calcium cyanide is the inorganic compound with the formula Ca(CN)2 . It is the calcium salt derived from hydrocyanic acid . It is a white solid, although the pure material is rarely encountered. It hydrolyses readily (even in moist air ) to release hydrogen cyanide and is very toxic .[3]
Preparation Solutions of calcium cyanide can be prepared by treating calcium hydroxide with hydrogen cyanide . Solid calcium cyanide is produced commercially by heating calcium cyanamide with sodium chloride . The reaction is incomplete. The product is only of 50% purity, other components being sodium cyanide, calcium cyanamide , and carbon. Because of the carbon impurity, the solid is black, hence material is often called black cyanide .[3]
Reactivity At temperatures around 600 °C, calcium cyanide converts to calcium cyanamide:[4] [5]
Ca(CN)2 → CaCN2 + C It is suspected that this reaction is one step in the conversion of calcium carbide with nitrogen gas. The ratio of calcium cyanide to calcium cyanamide is sensitive to the presence of alkali metal halides, such as sodium chloride.
Calcium cyanide hydrolyzes upon acidification to form hydrogen cyanide :
Ca(CN)2 + 2 H+ → Ca2+ + 2 HCN Calcium cyanide reacts with ammonium carbonate to give produce ammonium cyanide :
Ca(CN)2 + (NH4 )2 CO3 → 2 NH4 CN + CaCO3 Uses Calcium cyanide is used almost exclusively in the mining industry. It serves as an inexpensive source of cyanide in many leaching or vat operation to obtain precious metals such as gold and silver from their ores.[3] [6]
Safety Like other cyanide salts, this compound is highly toxic and its use is strictly regulated.
References External links
Salts and covalent derivatives of the
cyanide ion
HCN
He
LiCN
<style data-mw-deduplicate="TemplateStyles:r1123817410">'"`UNIQ--templatestyles-00000031-QINU`"'</style><span class="chemf nowrap">Be(CN)<sub class="template-chem2-sub">2</sub></span>
B(CN)<sub>3</sub>
C(CN)4 C2 (CN)2
NH4 CN ONCN O2 NCN N3 CN OCN− -NCO FCN
Ne
NaCN
Mg(CN)2 Al(CN)3 <link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Si(CN)<sub class="template-chem2-sub">4</sub></span>(CH3 )3 SiCN
P(CN)3 SCN− -NCS (SCN)2 S(CN)2 ClCN
Ar
KCN
Ca(CN)2
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Sc(CN)<sub class="template-chem2-sub">3</sub></span>
Ti
V
Cr(CN)6 3−
Mn
Fe(CN)2 Fe(CN)6 4− Fe(CN)6 3−
Co(CN)2 Ni(CN)2 Ni(CN)4 2− CuCN
Zn(CN)2 Ga(CN)3 <link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Ge(CN)<sub class="template-chem2-sub">2</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">As(CN)<sub class="template-chem2-sub">3</sub></span>(CH3 )2 AsCN (C6 H5 )2 AsCN
SeCN− BrCN
Kr
RbCN
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Sr(CN)<sub class="template-chem2-sub">2</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Y(CN)<sub class="template-chem2-sub">3</sub></span>
Zr
Nb
Mo(CN)8 4−
Tc
Ru
Rh
Pd(CN)2 AgCN
Cd(CN)2 <link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">In(CN)<sub class="template-chem2-sub">3</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Sn(CN)<sub class="template-chem2-sub">2</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Sb(CN)<sub class="template-chem2-sub">3</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Te(CN)<sub class="template-chem2-sub">2</sub></span>
ICN
Xe
CsCN
Ba(CN)2 *
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Lu(CN)<sub class="template-chem2-sub">3</sub></span>
Hf
Ta
W(CN)<sub>8</sub><sup>4−</sup>
Re
Os
Ir
Pt(CN)4 2- AuCN Au(CN)2 -
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Hg<sub class="template-chem2-sub">2</sub>(CN)<sub class="template-chem2-sub">2</sub></span>Hg(CN)2
TlCN
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Pb(CN)<sub class="template-chem2-sub">2</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Bi(CN)<sub class="template-chem2-sub">3</sub></span>
Po
At
Rn
Fr
Ra
**
Lr
Rf
Db
Sg
Bh
Hs
Mt
Ds
Rg
Cn
Nh
Fl
Mc
Lv
Ts
Og
*
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">La(CN)<sub class="template-chem2-sub">3</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Ce(CN)<sub class="template-chem2-sub">3</sub></span>
Pr
Nd
Pm
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Sm(CN)<sub class="template-chem2-sub">3</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Eu(CN)<sub class="template-chem2-sub">3</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Gd(CN)<sub class="template-chem2-sub">3</sub></span>
Tb
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Dy(CN)<sub class="template-chem2-sub">3</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Ho(CN)<sub class="template-chem2-sub">3</sub></span>
Er
Tm
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Yb(CN)<sub class="template-chem2-sub">3</sub></span>
**
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Ac(CN)<sub class="template-chem2-sub">3</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Th(CN)<sub class="template-chem2-sub">4</sub></span>
Pa
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">UO<sub class="template-chem2-sub">2</sub>(CN)<sub class="template-chem2-sub">2</sub></span>
Np
Pu
Am
Cm
Bk
Cf
Es
Fm
Md
No
Hydrogen & halogens Chalcogens Pnictogens Group 13 & 14 Trans metals Organics
This page was last edited on 8 November 2023, at 08:09