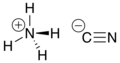Chemical compound
Ammonium cyanide is an unstable inorganic compound with the formula NH4 CN.
YouTube Encyclopedic
1 / 5
Views: 1 221
4 845
1 539
891
424
How to Write the Formula for Ammonium cyanide
Paradimethylaminobenzaldehyde
How to Write the Formula for Aluminum cyanide
The organic compound prepared by wholer from an inorgnic compound called ammonium cyanate was:
How to Write the Formula for Copper (II) cyanide
Uses Ammonium cyanide is generally used in organic synthesis. Being unstable, it is not shipped or sold commercially.
Preparation Ammonium cyanide is prepared in solution by bubbling hydrogen cyanide into aqueous ammonia at a low temperature
HCN + NH3 (aq) → NH4 CN (aq) It may be prepared by the reaction of calcium cyanide and ammonium carbonate :
Ca(CN)2 + (NH4 )2 CO3 → 2 NH4 CN + CaCO3 In dry state, ammonium cyanide is made by heating a mixture of potassium cyanide or potassium ferrocyanide with ammonium chloride and condensing the vapours into ammonium cyanide crystals:
KCN + NH4 Cl → NH4 CN + KCl Reactions Ammonium cyanide decomposes to ammonia and hydrogen cyanide, often forming a black polymer of hydrogen cyanide:[1]
NH4 CN → NH3 + HCN It undergoes salt metathesis reaction in solution with a number of metal salts to form metal–cyanide complexes.
Reaction with ketones and aldehydes yield aminonitriles, as in the first step of the Strecker amino acid synthesis :
NH4 CN + CH3 COCH3 → (CH3 )2 C(NH2 )CN + H2 O Toxicity The solid or its solution is highly toxic. Ingestion can cause death. Exposure to the solid can be harmful as it decomposes to highly toxic hydrogen cyanide and ammonia.
Chemical analysis Ammonium cyanide may be analyzed by heating the salt and trapping the decomposed products: hydrogen cyanide and ammonia in water at low temperatures. The aqueous solution is analyzed for cyanide ion by silver nitrate titrimetric method or an ion-selective
electrode method, and ammonia is measured by titration or electrode technique.
Notes
^ Matthews, Clifford N (1991). "Hydrogen cyanide polymerization: A preferred cosmochemical pathway". Bioastronomy: The Search for Extraterrestrial Life—The Exploration Broadens . Lecture Notes in Physics. Vol. 390. pp. 85–87. doi :10.1007/3-540-54752-5_195 . ISBN 978-3-540-54752-5
References A. F. Wells, Structural Inorganic Chemistry , 5th ed., Oxford University Press, Oxford, UK, 1984.
Inorganic salts
Organic salts
Salts and covalent derivatives of the
cyanide ion
HCN
He
LiCN
<style data-mw-deduplicate="TemplateStyles:r1123817410">'"`UNIQ--templatestyles-00000024-QINU`"'</style><span class="chemf nowrap">Be(CN)<sub class="template-chem2-sub">2</sub></span>
B(CN)<sub>3</sub>
C(CN)4 C2 (CN)2
NH4 CN ONCN O2 NCN N3 CN OCN− -NCO FCN
Ne
NaCN
Mg(CN)2 Al(CN)3 <link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Si(CN)<sub class="template-chem2-sub">4</sub></span>(CH3 )3 SiCN
P(CN)3 SCN− -NCS (SCN)2 S(CN)2 ClCN
Ar
KCN
Ca(CN)2
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Sc(CN)<sub class="template-chem2-sub">3</sub></span>
Ti
V
Cr(CN)6 3−
Mn
Fe(CN)2 Fe(CN)6 4− Fe(CN)6 3−
Co(CN)2 Ni(CN)2 Ni(CN)4 2− CuCN
Zn(CN)2 Ga(CN)3 <link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Ge(CN)<sub class="template-chem2-sub">2</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">As(CN)<sub class="template-chem2-sub">3</sub></span>(CH3 )2 AsCN (C6 H5 )2 AsCN
SeCN− BrCN
Kr
RbCN
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Sr(CN)<sub class="template-chem2-sub">2</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Y(CN)<sub class="template-chem2-sub">3</sub></span>
Zr
Nb
Mo(CN)8 4−
Tc
Ru
Rh
Pd(CN)2 AgCN
Cd(CN)2 <link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">In(CN)<sub class="template-chem2-sub">3</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Sn(CN)<sub class="template-chem2-sub">2</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Sb(CN)<sub class="template-chem2-sub">3</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Te(CN)<sub class="template-chem2-sub">2</sub></span>
ICN
Xe
CsCN
Ba(CN)2 *
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Lu(CN)<sub class="template-chem2-sub">3</sub></span>
Hf
Ta
W(CN)<sub>8</sub><sup>4−</sup>
Re
Os
Ir
Pt(CN)4 2- AuCN Au(CN)2 -
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Hg<sub class="template-chem2-sub">2</sub>(CN)<sub class="template-chem2-sub">2</sub></span>Hg(CN)2
TlCN
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Pb(CN)<sub class="template-chem2-sub">2</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Bi(CN)<sub class="template-chem2-sub">3</sub></span>
Po
At
Rn
Fr
Ra
**
Lr
Rf
Db
Sg
Bh
Hs
Mt
Ds
Rg
Cn
Nh
Fl
Mc
Lv
Ts
Og
*
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">La(CN)<sub class="template-chem2-sub">3</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Ce(CN)<sub class="template-chem2-sub">3</sub></span>
Pr
Nd
Pm
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Sm(CN)<sub class="template-chem2-sub">3</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Eu(CN)<sub class="template-chem2-sub">3</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Gd(CN)<sub class="template-chem2-sub">3</sub></span>
Tb
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Dy(CN)<sub class="template-chem2-sub">3</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Ho(CN)<sub class="template-chem2-sub">3</sub></span>
Er
Tm
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Yb(CN)<sub class="template-chem2-sub">3</sub></span>
**
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Ac(CN)<sub class="template-chem2-sub">3</sub></span>
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">Th(CN)<sub class="template-chem2-sub">4</sub></span>
Pa
<link href="mw-data:TemplateStyles:r1123817410" rel="mw-deduplicated-inline-style"/><span class="chemf nowrap">UO<sub class="template-chem2-sub">2</sub>(CN)<sub class="template-chem2-sub">2</sub></span>
Np
Pu
Am
Cm
Bk
Cf
Es
Fm
Md
No
This page was last edited on 25 January 2022, at 18:33