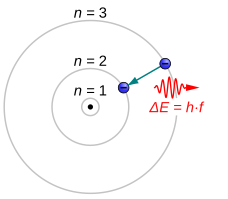
In atomic physics and chemistry, an atomic electron transition (also called an atomic transition, quantum jump, or quantum leap) is an electron changing from one energy level to another within an atom[1] or artificial atom.[2] The time scale of a quantum jump has not been measured experimentally. However, the Franck–Condon principle binds the upper limit of this parameter to the order of attoseconds.[3]
Electrons jumping to energy levels of smaller n emit electromagnetic radiation in the form of a photon. Electrons can also absorb passing photons, which drives a quantum jump to a level of higher n. The larger the energy separation between the electron's initial and final state, the shorter the photons' wavelength.[4]
YouTube Encyclopedic
-
1/3Views:1 144 14436 7889 259
-
Bohr Model of the Hydrogen Atom, Electron Transitions, Atomic Energy Levels, Lyman & Balmer Series
-
Electronic transitions and energy | AP Chemistry | Khan Academy
-
CHEMISTRY 101: Electron Transitions in a Hydrogen Atom
Transcription
History
Danish physicist Niels Bohr first theorized that electrons can perform quantum jumps in 1913.[5] Soon after, James Franck and Gustav Ludwig Hertz proved experimentally that atoms have quantized energy states.[6]
The observability of quantum jumps was predicted by Hans Dehmelt in 1975, and they were first observed using trapped ions of barium at University of Hamburg and mercury at NIST in 1986.[4]
Theory
An atom interacts with the oscillating electric field:
-
(1)
with amplitude , angular frequency , and polarization vector .[7] Note that the actual phase is . However, in many cases, the variation of is small over the atom (or equivalently, the radiation wavelength is much greater than the size of an atom) and this term can be ignored. This is called the dipole approximation. The atom can also interact with the oscillating magnetic field produced by the radiation, although much more weakly.
The Hamiltonian for this interaction, analogous to the energy of a classical dipole in an electric field, is . The stimulated transition rate can be calculated using time-dependent perturbation theory; however, the result can be summarized using Fermi's golden rule:
Recent discoveries
In 2019, it was demonstrated in an experiment with a superconducting artificial atom consisting of two strongly-hybridized transmon qubits placed inside a readout resonator cavity at 15 mK, that the evolution of some jumps is continuous, coherent, deterministic, and reversible.[8] On the other hand, other quantum jumps are inherently unpredictable.[9]
See also
References
- ^ Schombert, James. "Quantum physics" University of Oregon Department of Physics
- ^ Vijay, R; Slichter, D. H; Siddiqi, I (2011). "Observation of Quantum Jumps in a Superconducting Artificial Atom". Physical Review Letters. 106 (11): 110502. arXiv:1009.2969. Bibcode:2011PhRvL.106k0502V. doi:10.1103/PhysRevLett.106.110502. PMID 21469850. S2CID 35070320.
- ^ de la Peña, L.; Cetto, A. M.; Valdés-Hernández, A. (December 4, 2020). "How fast is a quantum jump?". Physics Letters A. 384 (34): 126880. arXiv:2009.02426. Bibcode:2020PhLA..38426880D. doi:10.1016/j.physleta.2020.126880. ISSN 0375-9601.
- ^ a b Itano, W. M.; Bergquist, J. C.; Wineland, D. J. (2015). "Early observations of macroscopic quantum jumps in single atoms" (PDF). International Journal of Mass Spectrometry. 377: 403. Bibcode:2015IJMSp.377..403I. doi:10.1016/j.ijms.2014.07.005.
- ^ Gleick, James (October 21, 1986). "PHYSICISTS FINALLY GET TO SEE QUANTUM JUMP WITH OWN EYES". The New York Times. ISSN 0362-4331. Retrieved December 6, 2021.
- ^ "Franck-Hertz experiment | physics | Britannica". www.britannica.com. Retrieved December 6, 2021.
- ^ Foot, CJ (2004). Atomic Physics. Oxford University Press. ISBN 978-0-19-850696-6.
- ^ Minev, Z. K.; Mundhada, S. O.; Shankar, S.; Reinhold, P.; Gutiérrez-Jáuregui, R.; Schoelkopf, R. J..; Mirrahimi, M.; Carmichael, H. J.; Devoret, M. H. (June 3, 2019). "To catch and reverse a quantum jump mid-flight". Nature. 570 (7760): 200–204. arXiv:1803.00545. Bibcode:2019Natur.570..200M. doi:10.1038/s41586-019-1287-z. PMID 31160725. S2CID 3739562.
- ^ Snizhko, Kyrylo; Kumar, Parveen; Romito, Alessandro (September 29, 2020). "Quantum Zeno effect appears in stages". Physical Review Research. 2 (3): 033512. arXiv:2003.10476. Bibcode:2020PhRvR...2c3512S. doi:10.1103/PhysRevResearch.2.033512. S2CID 214623209.
External links
- Schrödinger, Erwin (August 1952). "Are there quantum jumps? Part I" (PDF). The British Journal for the Philosophy of Science. 3 (10): 109–123. doi:10.1093/bjps/iii.10.109. Part 2
- "There are no quantum jumps, nor are there particles!" by H. D. Zeh, Physics Letters A172, 189 (1993).
- Ball, Philip (June 5, 2019). "Quantum Leaps, Long Assumed to Be Instantaneous, Take Time". Quanta Magazine. Retrieved June 6, 2019.
- "Surface plasmon at a metal-dielectric interface with an epsilon-near-zero transition layer" by Kevin Roccapriore et al., Physical Review B 103, L161404 (2021).








