

In chemistry, a radical, also known as a free radical, is an atom, molecule, or ion that has at least one unpaired valence electron.[1][2] With some exceptions, these unpaired electrons make radicals highly chemically reactive. Many radicals spontaneously dimerize. Most organic radicals have short lifetimes.
A notable example of a radical is the hydroxyl radical (HO·), a molecule that has one unpaired electron on the oxygen atom. Two other examples are triplet oxygen and triplet carbene (꞉CH
2) which have two unpaired electrons.
Radicals may be generated in a number of ways, but typical methods involve redox reactions, Ionizing radiation, heat, electrical discharges, and electrolysis are known to produce radicals. Radicals are intermediates in many chemical reactions, more so than is apparent from the balanced equations.
Radicals are important in combustion, atmospheric chemistry, polymerization, plasma chemistry, biochemistry, and many other chemical processes. A majority of natural products are generated by radical-generating enzymes. In living organisms, the radicals superoxide and nitric oxide and their reaction products regulate many processes, such as control of vascular tone and thus blood pressure. They also play a key role in the intermediary metabolism of various biological compounds. Such radicals can even be messengers in a process dubbed redox signaling. A radical may be trapped within a solvent cage or be otherwise bound.
YouTube Encyclopedic
-
1/5Views:858 80998 945151 177109 17083 117
-
Free radical reactions | Substitution and elimination reactions | Organic chemistry | Khan Academy
-
Initiation, Propagation, Termination - 3 Steps of Radical Reactions
-
Free Radical Reactions
-
Free Radicals
-
Radical Reactions & Hammond's Postulate: Crash Course Organic Chemistry #19
Transcription
Let's think about what type of reaction we might be able to get going if we had some methane and some molecular chlorine. So if we just let this be and we didn't heat it up or put in any UV light into this reaction, pretty much nothing will happen. Both of these molecules are reasonably happy being the way they are. But if we were to add heat into it, if we were to start making all the atoms and molecules vibrate more and bump into each other more, or we were to add energy in the form of UV light, what we could start doing is breaking some of these chlorine-chlorine bonds. Out of all of the bonds here, those are the weakest. That would be the most susceptible to breakage. So let's say we were to add some heat, what would happen? So let's see. Let me draw the valence electrons of each of these chlorines. This chlorine has one, two, three, four, five, six, seven valence electrons, and this chlorine over here has one, two, three, four, five, six, seven valence electrons. Now, when you add heat to this reaction, enough for these guys to vibrant away from each other, for this bond to break, what's going to happen, and we haven't drawn an arrow like this just yet, but what's going to happen is that each of these chlorines, this bond is going to break. Each of these chlorines are just going to take their part of the bond. So this guy on the left, he's just going to take his electron. And notice, I draw it with this half arrow. It looks like a fish hook. It's just half an arrowhead. This means that this electron is just going to go back to this chlorine, and this other magenta electron is going to go back to the right chlorine, so we can draw it like that. If it was up to me, I would have drawn it more like this. I would have drawn it more like this to show that that electron just goes back to the chlorine, but the convention shows that you can show that half of the bond is going back to the entire atom. Now, after this happens, what will everything look like? Well, we're still going to have our methane here. It hasn't really reacted. So we still have our methane. Let me draw it a little bit. So we still have our methane here. And all that's happened is, because we've put energy into the system, we've been able to break this bond. The molecular chlorine has broken up into two chlorine atoms. So we have the one on the left over here, and then we have the one on the right. And let me draw the left's valence electrons. It has one, two, three, four, five, six, seven. I just flipped it over so that the lone electron is on the left-hand side right here. And then you have the guy on the right. He has one, two, three, four, five, six, seven valence electrons. Now that each of these guys have an unpaired electron, they're actually very, very, very reactive. And we actually call any molecule that has an unpaired electron and is very reactive a free radical. So both of these guys now are free radicals. And actually, the whole topic of this video is free radical reactions. Both of these guys are free radicals. And you've probably heard the word free radical before. In the context of nutrition, that you don't want free radicals running around. And it's the exact same idea. It's not necessarily chlorine that they're talking about, but they're talking about molecules that have unpaired electrons. They'll react with some of your cell's machinery, maybe even with your DNA, maybe cause mutations that might lead to things like cancer. So that's why people think you shouldn't have free radicals in your body. But as soon as we form these free radicals, in this step right here, where we put energy in the system to break this bond, we call this the initiation step. Let me put this. We used energy here. This was endothermic. We use energy. This right here is the initiation step. And what we're going to see in general with free radical reactions is you need some energy to get it started. But once it gets started, it kind of starts this chain reaction. And as one free radical reacts with something else, it creates another free radical, and that keeps propagating until really everything has reacted. And that's why these can be so dangerous or so bad for biological systems. So I've told you that they react a lot. So how will they react now? Well, this guy wants to form a pair with someone else. And maybe if he swipes by this methane in just the right way, with just enough energy, what will happen is he could take the hydrogen off of the carbon, and not just the proton, the entire hydrogen. He will form a bond with the hydrogen using the hydrogen's electrons, so they'll get together and they'll form a bond. The hydrogen will contribute one electron. Notice, I'm drawing the half-arrow again, so the hydrogen isn't giving away the electron to someone else. That would be a full arrow. The hydrogen is just contributing its electron to half of a bond. And then the carbon, the carbon would do the same. I'll do that in blue. So the carbon, this valence electron right here, could be contributed to half of a bond, and then they will bond, and this bond over here will break. And so the carbon over here on the left, this carbon over here will take back its electron. So what does it look like? What does everything look like after that's done? So our methane now, it's no longer methane. It is now, if you think about it-- so we have three hydrogens. It took its electron back. It is now a free radical. It now has an unpaired reactive electron. The hydrogen and this chlorine have bonded. So let me draw the chlorine. It has this electron right over here. It has the other six valence electrons: one, two, three, four, five, six. And we have the hydrogen with its pink electron that it's contributing to the bond. And so we have them bonded now. This chlorine is no longer a free radical, although this one out here is still a free radical. Let me copy and paste it. So it's hanging around. Copy and paste. And now, notice we had one free radical react, but it formed another free radical. That's why we call this a propagation step. So this right here is a propagation step. When one free radical reacts, it created another free radical. Now, what's that free radical likely to do? You might be tempted to say, hey, it's going to just react with that other chlorine, but think about it. These molecules, there's a gazillion of them in this solution, so the odds that this guy's going to react exactly with that other free radical is actually very low, especially early on in the reaction where most of the molecules are still either methane or molecular chlorine. So this guy is much more likely to bump into another molecular chlorine than he is to bump into one of these original free radicals that formed. So if he bumps into another molecular chlorine in just the right way-- so let me draw another molecular chlorine. So that's another molecular chlorine. And each of these one, two, three, four, five six, seven; one, two, three, four, five, six, seven. There is a bond here. If they bump in just the right way, this chlorine electron might get contributed, and this free unpaired electron will be contributed and then this CH3, I guess we could call it, this free radical, this carbon free radical, or this methyl free radical, will then form a bond with this chlorine. What's everything going to look like after that? Well, after that happens this is now bonded to a chlorine. It's now chloromethane. Let me draw it. So it's carbon, hydrogen, hydrogen, hydrogen. Now, it's bonded to a chlorine. Let me draw the electrons so we can keep track of everything. We have that magenta electron right over there. And then we have the chlorine with its one, two, three, four, five, six, seven valence electrons. They are now bonded. This is chloromethane. And now you have another free radical because this guy-- and I should have drawn it there. This guy, that bond was broken, so he gets back his electrons. So he's sitting over here. He is now a free radical. So this is another propagation step. And we still have that original free radical guy sitting out over here. So we keep forming more and more free radicals as this happens. Now, eventually we're going to start running out of methanes and we're going to start running out of the molecular chlorines. So they're going to be less likely to react and you're actually going to have more free radicals around. So once the concentration of free radicals gets high enough, then you might start to see them reacting with each other. So when the concentration of free radicals get high enough, you might see, instead of this step happening-- this will happen a long time until most of the free radicals or most of the non-free radicals disappear. But once we have a soup of mainly free radicals, you'll see things like this. You'll see the methyl free radical. So let me draw it like this. You'll see him maybe reacting with another methyl free radical, where they both contribute an electron to form a bond. And then, once the bond forms, you have ethane. I could just write as CH3, H3C. So you might have something like this. And so this type of a step where two free radicals kind of cancel each other out, this is a termination step because it's starting to lower the concentration of free radicals in the solution, but this is only once the concentration of free radicals becomes really high. You might also see some of the chlorines cancel out with each other again, so a chlorine free radical and another chlorine free radical. I'll only draw the unpaired electron. They can bond with each other and form molecular chlorine again. That again is a termination step. Or you could see something like the methyl free radical. Just for shorthand, I'll write it like this: H3C. The methyl free radical and a chlorine free radical might also just straight-up react and form chloromethane, And form H3C-Cl. So this will all happen once the concentration of free radicals gets really high. Now, another thing that might happen once this reaction proceeds, and we have a lot of the propagation steps, is that you might have a situation where you already have a chloromethane, so it looks like this. You already have a chloromethane. And once you have enough of these, it then becomes more likely that some free radical chlorine might be able to react with this thing, so it might actually add another chlorine to this molecule. And the way it would do it, this chlorine over here-- I'm just drawing the free electron pairs. It would form a bond with this hydrogen right over there. They would both contribute their electrons. And then the carbon would take back its electron. Notice, all of the half-arrows. You'd be left with-- the hydrogen and the chlorine would have bonded. And now, this guy's going to be a free radical, but he's going to be a chlorinated free radical. So it's going to look like this. He has a free electron over there: hydrogen, hydrogen. And then he might be able to react with another chlorine molecule. He contributes an electron. Maybe this guy contributes an electron. This guy-- I don't want to draw a full arrow-- he contributes an electron to a bond, and then this guy takes his electron back and becomes a free radical. And then we're left with what? We're left with a doubly chlorinated methane. So then we have Cl, Cl, and then a hydrogen and a hydrogen. And this could actually keep happening. As the concentration of these get higher, then it becomes more likely that this can react with another chlorine. Of course, this chlorine over here becomes another free radical. But the general idea here that I wanted to show you is that once a free radical reaction starts-- the first step requires some energy to break this chlorine-chlorine bond, but once it happens, these guys are highly reactive, will start reacting with other things, and as they react with other things, it causes more and more free radicals, so it starts this chain reaction. And actually, all in all, this required energy to occur. This step right here, this propagation step, it requires a little bit of energy, but it's almost neutral. It requires energy to break this bond, but it creates energy when this bond is formed. It still requires a little net energy. And then things like this start to become exothermic. And especially once you start getting to the termination steps, you start releasing a lot of energy. So actually, all in all, this reaction is actually going to release energy, but it needed some energy to get started.
Formation
Radicals are either (1) formed from spin-paired molecules or (2) from other radicals. Radicals are formed from spin-paired molecules through homolysis of weak bonds or electron transfer, also known as reduction. Radicals are formed from other radicals through substitution, addition, and elimination reactions.
Radical formation from spin-paired molecules
Homolysis
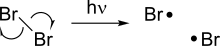
Homolysis makes two new radicals from a spin-paired molecule by breaking a covalent bond, leaving each of the fragments with one of the electrons in the bond.[3] Because breaking a chemical bond requires energy, homolysis occurs under the addition of heat or light. The bond dissociation energy associated with homolysis depends on the stability of a given compound, and some weak bonds are able to homolyze at relatively lower temperatures.
Some homolysis reactions are particularly important because they serve as an initiator for other radical reactions. One such example is the homolysis of halogens, which occurs under light and serves as the driving force for radical halogenation reactions.
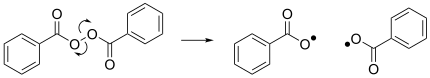
Another notable reaction is the homolysis of dibenzoyl peroxide, which results in the formation of two benzoyloxy radicals and acts as an initiator for many radical reactions.[4]
Reduction

Classically radicals form by one-electron reductions. Typically one-electron reduced organic compounds are unstable. Stability is conferred to the radical anion when the charge can be delocalized. Examples include alkali metal naphthenides, anthracenides, and ketyls.
Radical formation from other radicals
Abstraction

Hydrogen abstraction generates radicals. To achieve this reaction, the C-H bond of the H-atom donor must weak, which is rarely the case in organic compounds. Allylic and especiall doubly allylic C-H bonds are prone to abstraction by O2. This reaction is the basis of drying oils, such as linoleic acid derivatives.
Addition
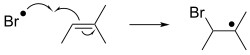
In free-radical additions, a radical adds to a spin-paired substrate. When applied to organic compounds, the reaction usually entails addition to an alkene. This addition generates a new radical, which can add to yet another alkene, etc. This behavior underpins radical polymerization, technology that produces many plastics.[5][6]
Elimination
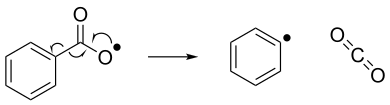
Radical elimination can be viewed as the reverse of radical addition. In radical elimination, an unstable radical compound breaks down into a spin-paired molecule and a new radical compound. Shown below is an example of a radical elimination reaction, where a benzoyloxy radical breaks down into a phenyl radical and a carbon dioxide molecule.[7]
Stability
Stability of organic radicals
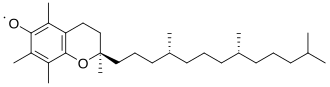
Although organic radicals are generally stable intrinsically (in isolation), practically speaking their existence is only transient because they tend to dimerize. Some are quite long-lived. Generally organic radicals are stabilized by any or all of these factors: presence of electronegativity, delocalization, and steric hindrance.[8] The compound 2,2,6,6-tetramethylpiperidinyloxyl illustrates the combination of all three factors. It is a commercially available solid that, aside from being magnetic, behaves like a normal organic compound.
Electronegativity
Organic radicals are inherently electron deficient thus the greater the electronegativity of the atom on which the unpaired electron resides the less stable the radical.[9] Between carbon, nitrogen, and oxygen, for example, carbon is the most stable and oxygen the least stable.
Electronegativity also factors into the stability of carbon atoms of different hybridizations. Greater s-character correlates to higher electronegativity of the carbon atom (due to the close proximity of s orbitals to the nucleus), and the greater the electronegativity the less stable a radical.[9] sp-hybridized carbons (50% s-character) form the least stable radicals compared to sp3-hybridized carbons (25% s-character) which form the most stable radicals.
Delocalization
The delocalization of electrons across the structure of a radical, also known as its ability to form one or more resonance structures, allows for the electron-deficiency to be spread over several atoms, minimizing instability. Delocalization usually occurs in the presence of electron-donating groups, such as hydroxyl groups (−OH), ethers (−OR), adjacent alkenes, and amines (−NH2 or −NR), or electron-withdrawing groups, such as C=O or C≡N.[3]

Delocalization effects can also be understood using molecular orbital theory as a lens, more specifically, by examining the intramolecular interaction of the unpaired electron with a donating group's pair of electrons or the empty π* orbital of an electron-withdrawing group in the form of a molecular orbital diagram. The HOMO of a radical is singly-occupied hence the orbital is aptly referred to as the SOMO, or the Singly-Occupied Molecular Orbital. For an electron-donating group, the SOMO interacts with the lower energy lone pair to form a new lower-energy filled bonding-orbital and a singly-filled new SOMO, higher in energy than the original. While the energy of the unpaired electron has increased, the decrease in energy of the lone pair forming the new bonding orbital outweighs the increase in energy of the new SOMO, resulting in a net decrease of the energy of the molecule. Therefore, electron-donating groups help stabilize radicals.

With a group that is instead electron-withdrawing, the SOMO then interacts with the empty π* orbital. There are no electrons occupying the higher energy orbital formed, while a new SOMO forms that is lower in energy. This results in a lower energy and higher stability of the radical species. Both donating groups and withdrawing groups stabilize radicals.
Another well-known albeit weaker form of delocalization is hyperconjugation. In radical chemistry, radicals are stabilized by hyperconjugation with adjacent alkyl groups. The donation of sigma (σ) C−H bonds into the partially empty radical orbitals helps to differentiate the stabilities of radicals on tertiary, secondary, and primary carbons. Tertiary carbon radicals have three σ C-H bonds that donate, secondary radicals only two, and primary radicals only one. Therefore, tertiary radicals are the most stable and primary radicals the least stable.
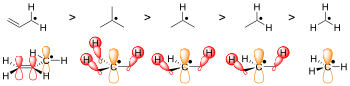
Steric hindrance

Most simply, the greater the steric hindrance the more difficult it is for reactions to take place, and the radical form is favored by default. For example, compare the hydrogen-abstracted form of N-hydroxypiperidine to the molecule TEMPO.[3] TEMPO, or (2,2,6,6-Tetramethylpiperidin-1-yl)oxyl, is too sterically hindered by the additional methyl groups to react making it stable enough to be sold commercially in its radical form. N-Hydroxypiperidine, however, does not have the four methyl groups to impede the way of a reacting molecule so the structure is unstable.[3]
Facile H-atom donors
The stability of many (or most) organic radicals is not indicated by their isolability but is manifested in their ability to function as donors of H•. This property reflects a weakened bond to hydrogen, usually O−H but sometimes N−H or C−H. This behavior is important because these H• donors serve as antioxidants in biology and in commerce. Illustrative is α-tocopherol (vitamin E). The tocopherol radical itself is insufficiently stable for isolation, but the parent molecule is a highly effective hydrogen-atom donor. The C−H bond is weakened in triphenylmethyl (trityl) derivatives.

Inorganic radicals
A large variety of inorganic radicals are stable and in fact isolable. Examples include most first-row transition metal complexes.
With regard to main group radicals, the most abundant radical in the universe is also the most abundant chemical in the universe, H•. Most main group radicals are not however isolable, despite their intrinsic stability. Hydrogen radicals for example combine eagerly to form H2. Nitric oxide (NO) is well known example of an isolable inorganic radical. Fremy's salt (Potassium nitrosodisulfonate, (KSO3)2NO) is a related example. Many thiazyl radicals are known, despite limited extent of π resonance stabilization.[10][11]
Many radicals can be envisioned as the products of breaking of covalent bonds by homolysis. The homolytic bond dissociation energies, usually abbreviated as "ΔH °" are a measure of bond strength. Splitting H2 into 2 H•, for example, requires a ΔH ° of +435 kJ/mol, while splitting Cl2 into two Cl• requires a ΔH ° of +243 kJ/mol. For weak bonds, homolysis can be induced thermally. Strong bonds require high energy photons or even flames to induce homolysis.
Diradicals
Diradicals are molecules containing two radical centers. Dioxygen (O2) is an important example of a stable diradical. Singlet oxygen, the lowest-energy non-radical state of dioxygen, is less stable than the diradical due to Hund's rule of maximum multiplicity. The relative stability of the oxygen diradical is primarily due to the spin-forbidden nature of the triplet-singlet transition required for it to grab electrons, i.e., "oxidize". The diradical state of oxygen also results in its paramagnetic character, which is demonstrated by its attraction to an external magnet.[12] Diradicals can also occur in metal-oxo complexes, lending themselves for studies of spin forbidden reactions in transition metal chemistry.[13] Carbenes in their triplet state can be viewed as diradicals centred on the same atom, while these are usually highly reactive persistent carbenes are known, with N-heterocyclic carbenes being the most common example.
Triplet carbenes and nitrenes are diradicals. Their chemical properties are distinct from the properties of their singlet analogues.
Occurrence of radicals
Combustion

A familiar radical reaction is combustion. The oxygen molecule is a stable diradical, best represented by •O–O•. Because spins of the electrons are parallel, this molecule is stable. While the ground state of oxygen is this unreactive spin-unpaired (triplet) diradical, an extremely reactive spin-paired (singlet) state is available. For combustion to occur, the energy barrier between these must be overcome. This barrier can be overcome by heat, requiring high temperatures. The triplet-singlet transition is also "forbidden". This presents an additional barrier to the reaction. It also means molecular oxygen is relatively unreactive at room temperature except in the presence of a catalytic heavy atom such as iron or copper.
Combustion consists of various radical chain reactions that the singlet radical can initiate. The flammability of a given material strongly depends on the concentration of radicals that must be obtained before initiation and propagation reactions dominate leading to combustion of the material. Once the combustible material has been consumed, termination reactions again dominate and the flame dies out. As indicated, promotion of propagation or termination reactions alters flammability. For example, because lead itself deactivates radicals in the gasoline-air mixture, tetraethyl lead was once commonly added to gasoline. This prevents the combustion from initiating in an uncontrolled manner or in unburnt residues (engine knocking) or premature ignition (preignition).
When a hydrocarbon is burned, a large number of different oxygen radicals are involved. Initially, hydroperoxyl radical (HOO•) are formed. These then react further to give organic hydroperoxides that break up into hydroxyl radicals (HO•).
Polymerization
Many polymerization reactions are initiated by radicals. Polymerization involves an initial radical adding to non-radical (usually an alkene) to give new radicals. This process is the basis of the radical chain reaction. The art of polymerization entails the method by which the initiating radical is introduced. For example, methyl methacrylate (MMA) can be polymerized to produce Poly(methyl methacrylate) (PMMA – Plexiglas or Perspex) via a repeating series of radical addition steps:
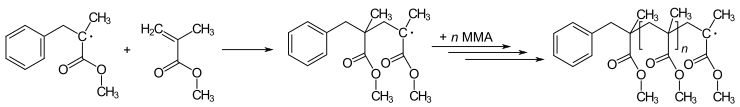
Newer radical polymerization methods are known as living radical polymerization. Variants include reversible addition-fragmentation chain transfer (RAFT) and atom transfer radical polymerization (ATRP).
Being a prevalent radical, O2 reacts with many organic compounds to generate radicals together with the hydroperoxide radical. Drying oils and alkyd paints harden due to radical crosslinking initiated by oxygen from the atmosphere.
Atmospheric radicals
The most common radical in the lower atmosphere is molecular dioxygen. Photodissociation of source molecules produces other radicals. In the lower atmosphere, important radical are produced by the photodissociation of nitrogen dioxide to an oxygen atom and nitric oxide (see eq. 1.1 below), which plays a key role in smog formation—and the photodissociation of ozone to give the excited oxygen atom O(1D) (see eq. 1.2 below). The net and return reactions are also shown (eq. 1.3 and eq. 1.4, respectively).
-
(eq. 1.1)
-
(eq. 1.2)
-
(eq. 1.3)
-
(eq. 1.4)
In the upper atmosphere, the photodissociation of normally unreactive chlorofluorocarbons (CFCs) by solar ultraviolet radiation is an important source of radicals (see eq. 1 below). These reactions give the chlorine radical, Cl•, which catalyzes the conversion of ozone to O2, thus facilitating ozone depletion (eq. 2.2–eq. 2.4 below).
-
(eq. 2.1)
-
(eq. 2.2)
-
(eq. 2.3)
-
(eq. 2.4)
-
(eq. 2.5)
Such reactions cause the depletion of the ozone layer, especially since the chlorine radical is free to engage in another reaction chain; consequently, the use of chlorofluorocarbons as refrigerants has been restricted.
In biology

Radicals play important roles in biology. Many of these are necessary for life, such as the intracellular killing of bacteria by phagocytic cells such as granulocytes and macrophages. Radicals are involved in cell signalling processes,[15] known as redox signaling. For example, radical attack of linoleic acid produces a series of 13-hydroxyoctadecadienoic acids and 9-hydroxyoctadecadienoic acids, which may act to regulate localized tissue inflammatory and/or healing responses, pain perception, and the proliferation of malignant cells. Radical attacks on arachidonic acid and docosahexaenoic acid produce a similar but broader array of signaling products.[16]
Radicals may also be involved in Parkinson's disease, senile and drug-induced deafness, schizophrenia, and Alzheimer's.[17] The classic free-radical syndrome, the iron-storage disease hemochromatosis, is typically associated with a constellation of free-radical-related symptoms including movement disorder, psychosis, skin pigmentary melanin abnormalities, deafness, arthritis, and diabetes mellitus. The free-radical theory of aging proposes that radicals underlie the aging process itself. Similarly, the process of mitohormesis suggests that repeated exposure to radicals may extend life span.
Because radicals are necessary for life, the body has a number of mechanisms to minimize radical-induced damage and to repair damage that occurs, such as the enzymes superoxide dismutase, catalase, glutathione peroxidase and glutathione reductase. In addition, antioxidants play a key role in these defense mechanisms. These are often the three vitamins, vitamin A, vitamin C and vitamin E and polyphenol antioxidants. Furthermore, there is good evidence indicating that bilirubin and uric acid can act as antioxidants to help neutralize certain radicals. Bilirubin comes from the breakdown of red blood cells' contents, while uric acid is a breakdown product of purines. Too much bilirubin, though, can lead to jaundice, which could eventually damage the central nervous system, while too much uric acid causes gout.[18]
Reactive oxygen species
Reactive oxygen species or ROS are species such as superoxide, hydrogen peroxide, and hydroxyl radical, commonly associated with cell damage. ROS form as a natural by-product of the normal metabolism of oxygen and have important roles in cell signaling. Two important oxygen-centered radicals are superoxide and hydroxyl radical. They derive from molecular oxygen under reducing conditions. However, because of their reactivity, these same radicals can participate in unwanted side reactions resulting in cell damage. Excessive amounts of these radicals can lead to cell injury and death, which may contribute to many diseases such as cancer, stroke, myocardial infarction, diabetes and major disorders.[19] Many forms of cancer are thought to be the result of reactions between radicals and DNA, potentially resulting in mutations that can adversely affect the cell cycle and potentially lead to malignancy.[20] Some of the symptoms of aging such as atherosclerosis are also attributed to radical induced oxidation of cholesterol to 7-ketocholesterol.[21] In addition radicals contribute to alcohol-induced liver damage, perhaps more than alcohol itself. Radicals produced by cigarette smoke are implicated in inactivation of alpha 1-antitrypsin in the lung. This process promotes the development of emphysema.
Oxybenzone has been found to form radicals in sunlight, and therefore may be associated with cell damage as well. This only occurred when it was combined with other ingredients commonly found in sunscreens, like titanium oxide and octyl methoxycinnamate.[22]
ROS attack the polyunsaturated fatty acid, linoleic acid, to form a series of 13-hydroxyoctadecadienoic acid and 9-hydroxyoctadecadienoic acid products that serve as signaling molecules that may trigger responses that counter the tissue injury which caused their formation. ROS attacks other polyunsaturated fatty acids, e.g. arachidonic acid and docosahexaenoic acid, to produce a similar series of signaling products.[23]
Reactive oxygen species are also used in controlled reactions involving singlet dioxygen known as type II photooxygenation reactions after Dexter energy transfer (triplet-triplet annihilation) from natural triplet dioxygen and triplet excited state of a photosensitizer. Typical chemical transformations with this singlet dioxygen species involve, among others, conversion of cellulosic biowaste into new poylmethine dyes.[24]
History and nomenclature

Until late in the 20th century the word "radical" was used in chemistry to indicate any connected group of atoms, such as a methyl group or a carboxyl, whether it was part of a larger molecule or a molecule on its own. A radical is often known as an R group. The qualifier "free" was then needed to specify the unbound case. Following recent nomenclature revisions, a part of a larger molecule is now called a functional group or substituent, and "radical" now implies "free". However, the old nomenclature may still appear in some books.
The term radical was already in use when the now obsolete radical theory was developed. Louis-Bernard Guyton de Morveau introduced the phrase "radical" in 1785 and the phrase was employed by Antoine Lavoisier in 1789 in his Traité Élémentaire de Chimie. A radical was then identified as the root base of certain acids (the Latin word "radix" meaning "root"). Historically, the term radical in radical theory was also used for bound parts of the molecule, especially when they remain unchanged in reactions. These are now called functional groups. For example, methyl alcohol was described as consisting of a methyl "radical" and a hydroxyl "radical". Neither are radicals in the modern chemical sense, as they are permanently bound to each other, and have no unpaired, reactive electrons; however, they can be observed as radicals in mass spectrometry when broken apart by irradiation with energetic electrons.
In a modern context the first organic (carbon–containing) radical identified was the triphenylmethyl radical, (C6H5)3C•. This species was discovered by Moses Gomberg in 1900. In 1933 Morris S. Kharasch and Frank Mayo proposed that free radicals were responsible for anti-Markovnikov addition of hydrogen bromide to allyl bromide.[25][26]
In most fields of chemistry, the historical definition of radicals contends that the molecules have nonzero electron spin. However, in fields including spectroscopy and astrochemistry, the definition is slightly different. Gerhard Herzberg, who won the Nobel prize for his research into the electron structure and geometry of radicals, suggested a looser definition of free radicals: "any transient (chemically unstable) species (atom, molecule, or ion)".[27] The main point of his suggestion is that there are many chemically unstable molecules that have zero spin, such as C2, C3, CH2 and so on. This definition is more convenient for discussions of transient chemical processes and astrochemistry; therefore researchers in these fields prefer to use this loose definition.[28]
Depiction in chemical reactions
In chemical equations, radicals are frequently denoted by a dot placed immediately to the right of the atomic symbol or molecular formula as follows:
Radical reaction mechanisms use single-headed arrows to depict the movement of single electrons:

The homolytic cleavage of the breaking bond is drawn with a "fish-hook" arrow to distinguish from the usual movement of two electrons depicted by a standard curly arrow. The second electron of the breaking bond also moves to pair up with the attacking radical electron.
Radicals also take part in radical addition and radical substitution as reactive intermediates. Chain reactions involving radicals can usually be divided into three distinct processes. These are initiation, propagation, and termination.
- Initiation reactions are those that result in a net increase in the number of radicals. They may involve the formation of radicals from stable species as in Reaction 1 above or they may involve reactions of radicals with stable species to form more radicals.
- Propagation reactions are those reactions involving radicals in which the total number of radicals remains the same.
- Termination reactions are those reactions resulting in a net decrease in the number of radicals. Typically two radicals combine to form a more stable species, for example:
- 2 Cl• → Cl2
See also
- Electron pair
- Globally Harmonized System of Classification and Labelling of Chemicals
- Hofmann–Löffler reaction
- Free radical research
References
- ^ IUPAC Gold Book radical (free radical) PDF Archived 2017-03-02 at the Wayback Machine
- ^ Hayyan, M.; Hashim, M.A.; Anjkut, I.M. (2016). "Superoxide Ion: Generation and Chemical Implications". Chem. Rev. 116 (5): 3029–85. doi:10.1021/acs.chemrev.5b00407. PMID 26875845.
- ^ a b c d Clayden, Jonathan; Greeves, Nick; Warren, Stuart G. (2012). Organic chemistry (2nd ed.). Oxford: Oxford University Press. ISBN 978-0-19-927029-3. OCLC 761379371.
- ^ "Diacyl Peroxides". polymerdatabase.com. Retrieved 2020-12-08.
- ^ Gridnev, Alexei A.; Ittel, Steven D. (2001). "Catalytic Chain Transfer in Free-Radical Polymerizations". Chemical Reviews. 101 (12): 3611–3660. doi:10.1021/cr9901236. PMID 11740917.
- ^ Monroe, Bruce M.; Weed, Gregory C. (1993). "Photoinitiators for free-radical-initiated photoimaging systems". Chemical Reviews. 93: 435–448. doi:10.1021/cr00017a019.
- ^ Su, Wei-Fang (2013), Su, Wei-Fang (ed.), "Radical Chain Polymerization", Principles of Polymer Design and Synthesis, Lecture Notes in Chemistry, vol. 82, Berlin, Heidelberg: Springer, pp. 137–183, doi:10.1007/978-3-642-38730-2_7, ISBN 978-3-642-38730-2
- ^ Griller, David; Ingold, Keith U. (1976). "Persistent carbon-centered radicals". Accounts of Chemical Research. 9: 13–19. doi:10.1021/ar50097a003.
- ^ a b Forrester, A.R. (1968). Organic Chemistry of Stable Free Radicals. London: Academic Press. pp. 1–6.
- ^ Oakley, Richard T. (1988). "Cyclic and Heterocyclic Thiazenes" (PDF). Progress in Inorganic Chemistry. Cyclic and Heterocyclic Thiazenes (section). Progress in Inorganic Chemistry. Vol. 36. pp. 299–391. doi:10.1002/9780470166376.ch4. ISBN 978-0-470-16637-6. Archived from the original (PDF) on 2015-09-23. Retrieved 2011-03-31.
- ^ Rawson, J; Banister, A; Lavender, I (1995). The Chemistry of Dithiadiazolylium and Dithiadiazolyl Rings. Advances in Heterocyclic Chemistry. Vol. 62. pp. 137–247. doi:10.1016/S0065-2725(08)60422-5. ISBN 978-0-12-020762-6.
- ^ However, paramagnetism does not necessarily imply radical character.
- ^ Linde, C.; Åkermark, B.; Norrby, P.-O.; Svensson, M. (1999). "Timing is Critical: Effect of Spin Changes on the Diastereoselectivity in Mn(Salen)-Catalyzed Epoxidation". Journal of the American Chemical Society. 121 (21): 5083–84. doi:10.1021/ja9809915.
- ^ Broderick, J.B.; Duffus, B.R.; Duschene, K.S.; Shepard, E.M. (2014). "Radical S-Adenosylmethionine Enzymes". Chemical Reviews. 114 (8): 4229–317. doi:10.1021/cr4004709. PMC 4002137. PMID 24476342.
- ^ Pacher P, Beckman JS, Liaudet L (2007). "Nitric oxide and peroxynitrite in health and disease". Physiol. Rev. 87 (1): 315–424. doi:10.1152/physrev.00029.2006. PMC 2248324. PMID 17237348.
- ^ Njie-Mbye, Ya Fatou; Kulkarni-Chitnis, Madhura; Opere, Catherine A.; Barrett, Aaron; Ohia, Sunny E. (2013). "Lipid peroxidation: pathophysiological and pharmacological implications in the eye". Frontiers in Physiology. 4: 366. doi:10.3389/fphys.2013.00366. PMC 3863722. PMID 24379787.
- ^ Floyd, R.A. (1999). "Neuroinflammatory processes are important in neurodegenerative diseases: An hypothesis to explain the increased formation of reactive oxygen and nitrogen species as major factors involved in neurodegenerative disease development". Free Radical Biology and Medicine. 26 (9–10): 1346–55. doi:10.1016/s0891-5849(98)00293-7. PMID 10381209.
- ^ An overview of the role of radicals in biology and of the use of electron spin resonance in their detection may be found in Rhodes C.J. (2000). Toxicology of the Human Environment – the critical role of free radicals. London: Taylor and Francis. ISBN 978-0-7484-0916-7.
- ^ Rajamani Karthikeyan; Manivasagam T; Anantharaman P; Balasubramanian T; Somasundaram ST (2011). "Chemopreventive effect of Padina boergesenii extracts on ferric nitrilotriacetate (Fe-NTA)-induced oxidative damage in Wistar rats". J. Appl. Phycol. 23 (2): 257–63. doi:10.1007/s10811-010-9564-0. S2CID 27537163.
- ^ Mukherjee, P.K.; Marcheselli, V.L.; Serhan, C.N.; Bazan, N.G. (2004). "Neuroprotecin D1: A docosahexanoic acid-derived docosatriene protects human retinal pigment epithelial cells from oxidative stress". Proceedings of the National Academy of Sciences of the USA. 101 (22): 8491–96. Bibcode:2004PNAS..101.8491M. doi:10.1073/pnas.0402531101. PMC 420421. PMID 15152078.
- ^ Lyons, MA; Brown, AJ (1999). "7-Ketocholesterol". Int. J. Biochem. Cell Biol. 31 (3–4): 369–75. doi:10.1016/s1357-2725(98)00123-x. PMID 10224662.
- ^ Serpone, N; Salinaro, A; Emeline, AV; Horikoshi, S; Hidaka, H; Zhao, JC (2002). "An in vitro systematic spectroscopic examination of the photostabilities of a random set of commercial sunscreen lotions and their chemical UVB/UVA active agents". Photochemical & Photobiological Sciences. 1 (12): 970–81. doi:10.1039/b206338g. PMID 12661594. S2CID 27248506.
- ^ Njie-Mbye, Ya Fatou; Kulkarni-Chitnis, Madhura; Opere, Catherine A.; Barrett, Aaron; Ohia, Sunny E. (2013). "Lipid peroxidation: pathophysiological and pharmacological implications in the eye". Frontiers in Physiology. 4: 366. doi:10.3389/fphys.2013.00366. PMC 3863722. PMID 24379787.
- ^ Desvals, Arthur; Fortino, Mariagrazia; Lefebvre, Corentin; Rogier, Johann; Michelin, Clément; Alioui, Samy; Rousset, Elodie; Pedone, Alfonso; Lemercier, Gilles; Hoffmann, Norbert (2022-05-16). "Synthesis and characterization of polymethine dyes carrying thiobarbituric and carboxylic acid moieties" (PDF). New Journal of Chemistry. 46 (19): 8971–8980. doi:10.1039/D2NJ00684G. ISSN 1369-9261. S2CID 248165785.
- ^ Kharasch, M. S. (1933). "The Peroxide Effect in the Addition of Reagents to Unsaturated Compounds. I. The Addition of Hydrogen Bromide to Allyl Bromide". Journal of the American Chemical Society. 55 (6): 2468–2496. doi:10.1021/ja01333a041.
- ^ Yan, M; Lo, JC; Edwards, JT; Baran, PS (2016). "Radicals: Reactive Intermediates with Translational Potential". J Am Chem Soc. 138 (39): 12692–12714. doi:10.1021/jacs.6b08856. PMC 5054485. PMID 27631602.
- ^ G. Herzberg (1971), "The spectra and structures of simple free radicals", ISBN 0-486-65821-X.
- ^ 28th International Symposium on Free Radicals Archived 2007-07-16 at the Wayback Machine.

![{\displaystyle {\ce {NO2 ->[h \nu] NO + O}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/5da627b27ee3e5b4dc45b156c54a91e1d79ffbd1)

![{\displaystyle {\ce {NO2 + O2 ->[h \nu] NO + O3}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/eae7fc4c91c01822b8db1423a88911a1eb56a171)

![{\displaystyle {\ce {CFCS->[h\nu ]Cl^{\bullet }}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/8bdfdc999e29261e922e3863cdfa3b4bf84fb5a2)

![{\displaystyle {\ce {O3 ->[h \nu] O + O2}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/3acd85b4a5f671e0a421576bf91c3352bec070c1)

![{\displaystyle {\ce {2O3 ->[h \nu] 3O2}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/afc621f0abb1c9cfc08b3cf82f43d9fc60a29c40)


