
Organotrifluoroborates are organoboron compounds that contain an anion with the general formula [RBF3]−. They can be thought of as protected boronic acids, or as adducts of carbanions and boron trifluoride. Organotrifluoroborates are tolerant of air and moisture[1] and are easy to handle and purify.[2] They are often used in organic synthesis as alternatives to boronic acids (RB(OH)2), boronate esters (RB(OR′)2), and organoboranes (R3B), particularly for Suzuki-Miyaura coupling.[3][4]
Structure
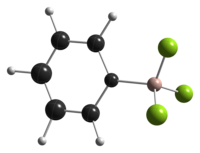 |

|
| thermal ellipsoid model of [PhBF3]−, as found in the crystal structure of K[PhBF3][5] |
space-filling packing diagram of the crystal structure of K[PhBF3][5] |
Synthesis
Boronic acids RB(OH)2 react with potassium bifluoride K[HF2] to form trifluoroborate salts K[RBF3].
Reactivity
Organotrifluoroborates are strong nucleophiles and react with electrophiles without transition-metal catalysts.[6]
Mechanism
The mechanism of organotrifluoroborate-based Suzuki-Miyaura coupling reactions has recently been investigated in detail. The organotrifluoroborate hydrolyses to the corresponding boronic acid in situ, so a boronic acid can be used in place of an organotrifluoroborate, as long as it is added slowly and carefully.[7][8]
References
- ^ "Alfa Aesar – Product Flyers & Technical Reviews – Organotrifluoroborates" (PDF).
- ^ "Sigma-Aldrich – Chemical Synthesis – Technology Spotlights – Organotrifluoroborates".
- ^ Darses, Sylvain; Genet, Jean-Pierre (2008). "Potassium Organotrifluoroborates: New Perspectives in Organic Synthesis". Chemical Reviews. 108 (1): 288–325. doi:10.1021/cr0509758. PMID 18095714.
- ^ Molander, Gary A.; Ellis, Noel (2007). "Organotrifluoroborates: Protected Boronic Acids That Expand the Versatility of the Suzuki Coupling Reaction". Accounts of Chemical Research. 40 (4): 275–286. doi:10.1021/ar050199q. PMID 17256882.
- ^ a b Conole, G.; Clough, A.; Whiting, A. (1995). "Potassium Trifluorophenylborate". Acta Crystallogr. C. 51 (6): 1056–1059. doi:10.1107/S0108270194014198.
- ^ Berionni, G.; Maji, B.; Knochel, P.; Mayr, H. (2012). "Nucleophilicity parameters for designing transition metal-free C–C bond forming reactions of organoboron compounds". Chemical Science. 3 (3): 878–882. doi:10.1039/C2SC00883A.
- ^ Butters, M.; Harvey, J. N.; Jover, J.; Lennox, A. J. J.; Lloyd-Jones, G. C.; Murray, P. M. (2010). "Aryl Trifluoroborates in Suzuki-Miyaura Coupling: The Roles of Endogenous Aryl Boronic Acid and Fluoride". Angew. Chem. Int. Ed. 49 (30): 5156–60. doi:10.1002/anie.201001522. PMID 20544767.
- ^ "Mechanism Of Suzuki-Miyaura Coupling Revealed". Chemical & Engineering News. Retrieved 2010-06-27.
