In chemistry, the descriptor geminal (from Latin gemini 'twins'[1]) refers to the relationship between two atoms or functional groups that are attached to the same atom. A geminal diol, for example, is a diol (a molecule that has two alcohol functional groups) attached to the same carbon atom, as in methanediol. Also the shortened prefix gem may be applied to a chemical name to denote this relationship, as in a gem-dibromide for "geminal dibromide".[citation needed]
The concept is important in many branches of chemistry, including synthesis and spectroscopy, because functional groups attached to the same atom often behave differently from when they are separated. Geminal diols, for example, are easily converted to ketones or aldehydes with loss of water.[2]
| Alkane | geminal | vicinal | isolated | |
| Methane | 
|
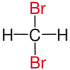
|
not existing | not existing |
| Ethane | 
|
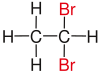
|

|
not existing |
| Propane | 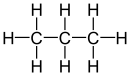
|
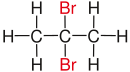
|

|
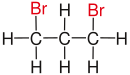
|
| Substituents on selected dibromoalkanes labeled red. | ||||
The related term vicinal refers to the relationship between two functional groups that are attached to adjacent atoms. The relative arrangement of two functional groups can also be described by the descriptors α and β.
YouTube Encyclopedic
-
1/3Views:82 02720 272918
-
Preparation of alkynes | Alkenes and Alkynes | Organic chemistry | Khan Academy
-
NMR Coupling Constants in Organic Chemistry
-
Geminal Dibromide with Sodium Amide Mechanism 007
Transcription
Let's look at two ways to prepare alkynes from alkyl halides. So here I have an alkyl halide. So this is a dihalide, and my two halogens are attached to one carbon. We call this a geminal dihalide. So this is going to be a geminal dihalide reacting with a very strong base, sodium amide. So this is going to give us an E2 elimination reaction. So we're going to get an E2 elimination reaction, and this E2 elimination reaction is actually going to occur twice. And we're going to end up with an alkyne as our final product. So let's take a look at the mechanism of our double E2 elimination of a geminal dihalide. So let's start with our dihalide over here. And this time we're going to put in all of our lone pairs of electrons on our halogen, like that. So let me go ahead and put all of those in there, and then I have two hydrogens on this carbon. OK. Sodium amide is a source of amide anions, which we saw in our previous video can function as a strong base. So a strong base means that a lone pair of electrons here on our nitrogen is going to take this proton. And these electrons, in here, are going to kick in to form a double bond at the same time these electrons kick off onto our halogen. So an E2 elimination mechanism. You can watch the previous videos on E2 elimination reactions for more details. So we're going to form ammonia as one of our products. And our other product is going to be carbon double-bonded to another carbon. And then we're going to still have our halogen down here. And over here, in the carbon on the right, we're still going to have a hydrogen, like that. So we're not quite to our alkyne yet. So we've done one E2 elimination reaction, and we're going to do one more. So we get another-- another amide anion comes along, and it's negatively charged. It's going to function as a base. It's going to take this proton this time. And these electrons are going to move in here to form our triple bond. And these electrons are going to kick off onto our halogen, like that. So that is going to finally form our alkyne here. So you always have to have your base in excess, if you're trying to do this. Let's look at a very similar reaction, a double E2 elimination. This time the halogens are not on the same carbon. So let's go ahead and draw the general reaction for this. We have two carbons right here, and we have two halogens right here. And then hydrogen, and then hydrogen. This time we have two halogens on adjacent carbons. So this is called vicinal dihalides. So let's go ahead and write that. So this is vicinal, and the one we did before was geminal. So a vicinal dihalide will react in a very similar way if you add a strong base like sodium amide and you use ammonia for your solvent. So you're going to form an alkyne once again. So you're going to get an alkyne. It's going to be via a double E2 elimination reaction again. Let's look at the mechanism. So let's start with our vicinal dihalide down here. So let's go ahead and put our halogens in there. Lone pairs of electrons on our halogens, like that. And then we have hydrogen, and we have hydrogen right here. So we have our amide anion, and once again, functions as a strong base. It's going to take a proton. So it's going to take this proton right here. These electrons are going to move in to form our double bond the same time these electrons kick off on to our halogen. So that's our first E2 elimination reaction. So let's just go ahead and write E2 here to remind us this is yet another E2 reaction. And let's go ahead and draw the product of that. So now we're going to have carbon double-bonded to another carbon. And then we're going to have a hydrogen right here. And then we're going to have our halogen up here, like that. And then we're going to have-- we need one more reaction to form our alkyne. We're going to get another E2 elimination reaction. So sodium amide-- another anion of sodium amide comes along. So let's go ahead and put in those lone pairs of electrons, like that. It's going to function as a base. Lone pair of electrons takes this proton. These electrons kick in here to form our triple bond at the same time our halogen leaves. And so we form our alkyne like that. So you can produce alkynes from either vicinal or geminal dihalides via a double E2 elimination reaction. Let's see how we could use this in a synthesis reaction. So let's go ahead and try to make something-- try to make an alkyne from an alkene. OK, so let's start with an alkene here. And I'll put some benzene rings on this guy here. So here's a benzene ring like that. Put in my lone pairs-- sorry, put in my bonds like that. And then I'm going to put a double bond right here. And then I'm going to put another benzene ring attached like that. So this is 1,2-Diphenylethylene. . And I'm going to react this alkene with bromine. And you could use a solvent like carbon tetrachloride or something like that. And we're reacting an alkene with a halogen. And we've seen this reaction before in the videos on reactions of alkenes. We're going to add two bromines across our double bond. So we're going to draw the product of this reaction. Our benzene rings aren't going to react as readily as our double bonds will. So let's go ahead and draw in our other benzene ring here, like that. And we know that we're going to add a bromine to either side of our double bond. So let's go ahead and add a bromine to either side of our double bond here. And we'll also have a hydrogen bonded to each one of these carbons. That hydrogen was originally there as well, over here on the left. And we form 1,2-Dibromo-1,2-diphenylethane here. And now we have a vicinal dihalide. So if we add a strong base to our vicinal dihalide we can prepare an alkyne from that. So if we add an excess of sodium amide in ammonia we know that we're going to get a double E2 elimination reaction. And those halogens are going to go away in our double E2 elimination reaction and form a triple bond. So we're going to form a triple bond. So those two carbons, the ones that form a triple bond, are these two carbons right here. So when you run through the mechanism, you're going to get an alkyne. And then, on either side of that alkyne, you're going to get a phenyl groups. So let's go ahead and draw in our benzene rings, like that. So it doesn't really matter how we draw our electrons, so we'll go ahead and do this. So this would be our product. So let's go ahead and put in those electrons. This will be diphenylacetylene. So you can synthesize alkynes from alkenes, or you could synthesize an alkyne from a dihalide. So this is one way to do it.
1H NMR spectroscopy
In 1H NMR spectroscopy, the coupling of two hydrogen atoms on the same carbon atom is called a geminal coupling. It occurs only when two hydrogen atoms on a methylene group differ stereochemically from each other. The geminal coupling constant is referred to as 2J since the hydrogen atoms couple through two bonds. Depending on the other substituents, the geminal coupling constant takes values between −23 and +42 Hz.[3][4]
Synthesis
The following example shows the conversion of a cyclohexyl methyl ketone to a gem-dichloride through a reaction with phosphorus pentachloride. This gem-dichloride can then be used to synthesize an alkyne.

References
- ^ "Definition of GEMINI". Merriam-Webster Dictionary. 27 January 2021. Retrieved 27 January 2021.
- ^ Peter Taylor (2002), Mechanism and synthesis, Book 10 of Molecular world. Open University, Royal Society of Chemistry; ISBN 0-85404-695-X. 368 pages
- ^ H. Günther: NMR-Spektroskopie; Grundlagen,Konzepte und Anwendungen der Protonen- und Kohlenstoff-13-Kernresonanzspektroskopie in der Chemie. 3. neubearbeitete und erweiterte Auflage, Georg Thieme Verlag, Stuttgart 1992, S. 103.
- ^ D. H. Williams, I. Fleming: Strukturaufklärung in der organischen Chemie; Eine Einführung in die spektroskopischen Methoden. 6. überarbeitete Auflage, Georg Thieme Verlag, Stuttgart 1991, S. 109.
