Gaseous signaling molecules are gaseous molecules that are either synthesized internally (endogenously) in the organism, tissue or cell or are received by the organism, tissue or cell from outside (say, from the atmosphere or hydrosphere, as in the case of oxygen) and that are used to transmit chemical signals which induce certain physiological or biochemical changes in the organism, tissue or cell. The term is applied to, for example, oxygen, carbon dioxide, sulfur dioxide, nitrous oxide, hydrogen cyanide, ammonia, methane, hydrogen, ethylene, etc.
Select gaseous signaling molecules behave as neurotransmitters and are called gasotransmitters. These include nitric oxide, carbon monoxide, and hydrogen sulfide.
Historically, the study of gases and physiological effects was categorized under factitious airs.
The biological roles of each of the gaseous signaling molecules are outlined below.
YouTube Encyclopedic
-
1/5Views:3 64235 9562 04414 422683 948
-
What are Ligands? 6 Types of Ligands or Signaling molecules in Cell Signaling
-
Cell to Cell Communication || Types of signaling
-
Signaling Part 1
-
TS/AP SENIOR INTER CHEMISTRY IPE II p-BLOCK ELEMENTS (2 MARKS) II MASS PHYSICS-MNR
-
Gas chromatography | Chemical processes | MCAT | Khan Academy
Transcription
Gasotransmitters
Gasotransmitters are a class of neurotransmitters. Only three gases are accepted to be classified as gasotransmitters including nitric oxide, carbon monoxide, and hydrogen sulfide.
Gaseous Signaling Molecules
Oxygen
Oxygen, O2, is an essential gaseous signaling molecule & biological messenger important in many physiological and pathological processes, acting via cellular gasoreceptor proteins and other signaling pathways.[1][2] The levels of O2 in cells or organisms must be tighly regulated to ensure normoxic and not uncontrolled hypoxic or anoxic or hyperoxic states. In mammals, specialized tissues such as carotid body sense O2 levels.
Carbon dioxide
Carbon dioxide, CO2, is one of the mediators of local autoregulation of blood supply. If its levels are high, the capillaries expand to allow a greater blood flow to that tissue.
Mosquitoes are attracted to humans by sensing the CO2 via gustatory receptors, a type of gasoreceptor.[3]
Although the body requires oxygen for metabolism, low oxygen levels normally do not stimulate breathing. Rather, breathing is stimulated by higher carbon dioxide levels.[4]
The respiratory centers try to maintain an arterial CO2 pressure of 40 mm Hg. With intentional hyperventilation, the CO2 content of arterial blood may be lowered to 10–20 mm Hg (the oxygen content of the blood is little affected), and the respiratory drive is diminished. This is why one can hold one's breath longer after hyperventilating than without hyperventilating. This carries the risk that unconsciousness may result before the need to breathe becomes overwhelming, which is why hyperventilation is particularly dangerous before free diving.
Nitric oxide
Nitric oxide, NO, is a key vertebrate biological messenger important in many physiological and pathological processes, acting, for instance, as a powerful vasodilator in humans (see Biological functions of nitric oxide). Mammalian cells have a specialized gasoreceptor soluble guanylyl cyclase that bind to NO and trigger NO-dependent cellular signaling.
Nitrous oxide
Nitrous oxide, N2O, in biological systems can be formed by an enzymatic or non-enzymatic reduction of nitric oxide.[5] In vitro studies have shown that endogenous nitrous oxide can be formed by the reaction between nitric oxide and thiol.[6] Some authors have shown that this process of NO reduction to N2O takes place in hepatocytes, specifically in their cytoplasm and mitochondria, and suggested that the N2O can possibly be produced in mammalian cells.[7] It is well known that N2O is produced by some bacteria during process called denitrification.[8]
In 1981, it was first suggested from clinical work with nitrous oxide (N2O) that a gas had a direct action at pharmacological receptors and thereby acted as a neurotransmitter.[9][10][11] In vitro experiments confirmed these observations[12] which were replicated at NIDA later.[13]
Apart from its direct[14][15] and indirect actions at opioid receptors,[16] it was also shown that N2O inhibits NMDA receptor-mediated activity and ionic currents and diminishes NMDA receptor-mediated excitotoxicity and neurodegeneration.[17] Nitrous oxide also inhibits methionine synthase and slows the conversion of homocysteine to methionine, increases homocysteine concentration and decreases methionine concentration. This effect was shown in lymphocyte cell cultures[18] and in human liver biopsy samples.[19]
Nitrous oxide does not bind as a ligand to the heme and does not react with thiol-containing proteins. Nevertheless, studies have shown that nitrous oxide can reversibly and non-covalently "insert" itself into the inner structures of some heme-containing proteins such as hemoglobin, myoglobin, cytochrome oxidase and alter their structure and function.[20] The ability of nitrous oxide to alter the structure and function of these proteins was demonstrated by shifts in infrared spectra of cysteine thiols of hemoglobin[21] and by partial and reversible inhibition of cytochrome oxidase.[22]
Endogenous nitrous oxide can possibly play a role in modulating endogenous opioid[23][24] and NMDA systerosclerosis, severe sepsis, severe malaria, or autoimmunity. Clinical tests involving humans have been performed, but the results have not yet been released.[25]
Carbon suboxide

Carbon suboxide, C3O2, can be produced in small amounts in any biochemical process that normally produces carbon monoxide, CO, for example, during heme oxidation by heme oxygenase-1. It can also be formed from malonic acid. It has been shown that carbon suboxide in an organism can quickly polymerize into macrocyclic polycarbon structures with the common formula (C3O2)n (mostly (C3O2)6 and (C3O2)8), and that those macrocyclic compounds are potent inhibitors of Na+/K+-ATP-ase and Ca-dependent ATP-ase, and have digoxin-like physiological properties and natriuretic and antihypertensive actions. Those macrocyclic carbon suboxide polymer compounds are thought to be endogenous digoxin-like regulators of Na+/K+-ATP-ases and Ca-dependent ATP-ases, and endogenous natriuretics and antihypertensives.[26][27][28] Other than that, some authors think also that those macrocyclic compounds of carbon suboxide can possibly diminish free radical formation and oxidative stress and play a role in endogenous anticancer protective mechanisms, for example in the retina.[29]
Sulfur dioxide
The role of sulfur dioxide, SO2, in mammalian biology is not well understood.[30] Sulfur dioxide blocks nerve signals from the pulmonary stretch receptors and abolishes the Hering–Breuer inflation reflex.
Sulfur dioxide plays a role in diminishing an experimental lung damage caused by oleic acid. Endogenous sulfur dioxide lowered lipid peroxidation, free radical formation, oxidative stress and inflammation during an experimental lung damage. Conversely, a successful lung damage caused a significant lowering of endogenous sulfur dioxide production, and an increase in lipid peroxidation, free radical formation, oxidative stress and inflammation. Moreover, blockade of an enzyme that produces endogenous SO2 significantly increased the amount of lung tissue damage in the experiment. Conversely, adding acetylcysteine or glutathione to the rat diet increased the amount of endogenous SO2 produced and decreased the lung damage, the free radical formation, oxidative stress, inflammation and apoptosis.[31]
Endogenous sulfur dioxide may play a role in regulating cardiac and blood vessel function, and aberrant or deficient sulfur dioxide metabolism can contribute to several different cardiovascular diseases, such as arterial hypertension, atherosclerosis, pulmonary arterial hypertension, stenocardia.[32]
In children with pulmonary arterial hypertension due to congenital heart diseases, the level of homocysteine is higher and the level of endogenous sulfur dioxide is lower than in normal control children. Moreover, these biochemical parameters strongly correlated to the severity of pulmonary arterial hypertension. Authors considered homocysteine to be one of useful biochemical markers of disease severity and sulfur dioxide metabolism to be one of potential therapeutic targets in those patients.[33]
Endogenous sulfur dioxide also lowers the proliferation rate of endothelial smooth muscle cells in blood vessels, via lowering the MAPK activity and activating adenylyl cyclase and protein kinase A.[34] Smooth muscle cell proliferation is one of important mechanisms of hypertensive remodeling of blood vessels and their stenosis, so it is an important pathogenetic mechanism in arterial hypertension and atherosclerosis.
Endogenous sulfur dioxide in low concentrations causes endothelium-dependent vasodilation. In higher concentrations it causes endothelium-independent vasodilation and has a negative inotropic effect on cardiac output function, thus effectively lowering blood pressure and myocardial oxygen consumption. The vasodilating effects of sulfur dioxide are mediated via ATP-dependent calcium channels and L-type ("dihydropyridine") calcium channels. Endogenous sulfur dioxide is also a potent antiinflammatory, antioxidant and cytoprotective agent. It lowers blood pressure and slows hypertensive remodeling of blood vessels, especially thickening of their intima. It also regulates lipid metabolism.[35]
Endogenous sulfur dioxide also diminishes myocardial damage, caused by isoproterenol adrenergic hyperstimulation, and strengthens the myocardial antioxidant defense reserve.[36]
Hydrogen cyanide
Some authors have shown that neurons can produce hydrogen cyanide, HCN, upon activation of their opioid receptors by endogenous or exogenous opioids. They have also shown that neuronal production of HCN activates NMDA receptors and plays a role in signal transduction between neuronal cells (neurotransmission). Moreover, increased endogenous neuronal HCN production under opioids was seemingly needed for adequate opioid analgesia, as analgesic action of opioids was attenuated by HCN scavengers. They considered endogenous HCN to be a neuromodulator.[37]
It was also shown that, while stimulating muscarinic cholinergic receptors in cultured pheochromocytoma cells increases HCN production, in a living organism (in vivo) muscarinic cholinergic stimulation actually decreases HCN production.[38]
Leukocytes generate HCN during phagocytosis.[37]
The vasodilatation, caused by sodium nitroprusside, has been shown to be mediated not only by NO generation, but also by endogenous cyanide generation, which adds not only toxicity, but also some additional antihypertensive efficacy compared to nitroglycerine and other non-cyanogenic nitrates which do not cause blood cyanide levels to rise.[39]
Ammonia
Ammonia, NH3, also plays a role in both normal and abnormal animal physiology. It is biosynthesised through normal amino acid metabolism, but is toxic in high concentrations.[40] The liver converts ammonia to urea through a series of reactions known as the urea cycle. Liver dysfunction, such as that seen in cirrhosis, may lead to elevated amounts of ammonia in the blood (hyperammonemia). Likewise, defects in the enzymes responsible for the urea cycle, such as ornithine transcarbamylase, lead to hyperammonemia. Hyperammonemia contributes to the confusion and coma of hepatic encephalopathy, as well as the neurologic disease common in people with urea cycle defects and organic acidurias.[41]
Ammonia is important for normal animal acid/base balance. After formation of ammonium from glutamine, α-ketoglutarate may be degraded to produce two molecules of bicarbonate, which are then available as buffers for dietary acids. Ammonium is excreted in the urine, resulting in net acid loss. Ammonia may itself diffuse across the renal tubules, combine with a hydrogen ion, and thus allow for further acid excretion.[42]
Methane
Some authors have shown that endogenous methane, CH4, is produced not only by the intestinal flora and then absorbed into the blood, but also is produced - in small amounts - by eukaryotic cells (during process of lipid peroxidation). And they have also shown that the endogenous methane production rises during an experimental mitochondrial hypoxia, for example, sodium azide intoxication. They thought that methane could be one of intercellular signals of hypoxia and stress.[43]
Other authors have shown that cellular methane production also rises during sepsis or bacterial endotoxemia, including an experimental imitation of endotoxemia by lipopolysaccharide (LPS) administration.[44]
Some other researchers have shown that methane, produced by the intestinal flora, is not fully "biologically neutral" to the intestine, and it participates in the normal physiologic regulation of peristalsis. And its excess causes not only belching, flatulence and belly pain, but also functional constipation.[45]
Ethylene

Ethylene, H2C=CH2, serves as a hormone in plants.[46] It acts at trace levels throughout the life of the plant by stimulating or regulating the ripening of fruit, the opening of flowers, and the abscission (or shedding) of leaves. Commercial ripening rooms use "catalytic generators" to make ethylene gas from a liquid supply of ethanol. Typically, a gassing level of 500 to 2,000 ppm is used, for 24 to 48 hours. Care must be taken to control carbon dioxide levels in ripening rooms when gassing, as high temperature ripening (20 °C; 68 °F) has been seen to produce CO2 levels of 10% in 24 hours.[47]
Ethylene has been used since the ancient Egyptians, who would gash figs in order to stimulate ripening (wounding stimulates ethylene production by plant tissues). The ancient Chinese would burn incense in closed rooms to enhance the ripening of pears. In 1864, it was discovered that gas leaks from street lights led to stunting of growth, twisting of plants, and abnormal thickening of stems.[46] In 1901, a Russian scientist named Dimitry Neljubow showed that the active component was ethylene.[48] Sarah Doubt discovered that ethylene stimulated abscission in 1917.[49] It wasn't until 1934 that Gane reported that plants synthesize ethylene.[50] In 1935, Crocker proposed that ethylene was the plant hormone responsible for fruit ripening as well as senescence of vegetative tissues.[51]
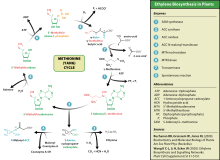
Ethylene is produced from essentially all parts of higher plants, including leaves, stems, roots, flowers, fruits, tubers, and seeds. Ethylene production is regulated by a variety of developmental and environmental factors. During the life of the plant, ethylene production is induced during certain stages of growth such as germination, ripening of fruits, abscission of leaves, and senescence of flowers. Ethylene production can also be induced by a variety of external aspects such as mechanical wounding, environmental stresses, and certain chemicals including auxin and other regulators.[52]
Ethylene is biosynthesized from the amino acid methionine to S-adenosyl-L-methionine (SAM, also called Adomet) by the enzyme Met Adenosyltransferase. SAM is then converted to 1-aminocyclopropane-1-carboxylic acid (ACC) by the enzyme ACC synthase (ACS). The activity of ACS determines the rate of ethylene production, therefore regulation of this enzyme is key for the ethylene biosynthesis. The final step requires oxygen and involves the action of the enzyme ACC-oxidase (ACO), formerly known as the ethylene forming enzyme (EFE). Ethylene biosynthesis can be induced by endogenous or exogenous ethylene. ACC synthesis increases with high levels of auxins, especially indole acetic acid (IAA) and cytokinins.
Ethylene is perceived by a family of five transmembrane protein dimers such as the ETR1 gasoreceptor protein in Arabidopsis. The gene encoding an ethylene receptor[which?] has been cloned in Arabidopsis thaliana and then in tomato.[citation needed] Ethylene receptors are encoded by multiple genes in the Arabidopsis and tomato genomes. Mutations in any of the gene family, which comprises five receptors in Arabidopsis and at least six in tomato, can lead to insensitivity to ethylene.[53] DNA sequences for ethylene receptors have also been identified in many other plant species and an ethylene binding protein has even been identified in Cyanobacteria.[46]
Environmental cues such as flooding, drought, chilling, wounding, and pathogen attack can induce ethylene formation in plants. In flooding, roots suffer from lack of oxygen, or anoxia, which leads to the synthesis of 1-aminocyclopropane-1-carboxylic acid (ACC). ACC is transported upwards in the plant and then oxidized in leaves. The ethylene produced causes nastic movements (epinasty) of the leaves, perhaps helping the plant to lose water.[54]
Ethylene in plant induces such responses:
- Seedling triple response, thickening and shortening of hypocotyl with pronounced apical hook.
- In pollination, when the pollen reaches the stigma, the precursor of the ethene, ACC, is secreted to the petal, the ACC releases ethylene with ACC oxidase.
- Stimulates leaf and flower senescence
- Stimulates senescence of mature xylem cells in preparation for plant use
- Induces leaf abscission
- Induces seed germination
- Induces root hair growth [55]— increasing the efficiency of water and mineral absorption through rhizosheath formation [56][57]
- Induces the growth of adventitious roots during flooding [58][59]
- Stimulates survival under low-oxygen conditions (hypoxia) in submerged plant tissues [60][61][62][63]
- Stimulates epinasty — leaf petiole grows out, leaf hangs down and curls into itself
- Stimulates fruit ripening[64]
- Induces a climacteric rise in respiration in some fruit which causes a release of additional ethylene.
- Affects gravitropism
- Inhibits root growth in response to soil compaction,[65][66] shade [67][68] and flooding [63]
- Stimulates nutational bending
- Inhibits stem growth and stimulates stem and cell broadening and lateral branch growth outside of seedling stage (see Hyponastic response)
- Interference with auxin transport (with high auxin concentrations)
- Inhibits shoot growth and stomatal closing except in some water plants or habitually flooded ones such as some rice varieties, where the opposite occurs (conserving CO
2 and O
2) - Induces flowering in pineapples
- Inhibits short day induced flower initiation in Pharbitus nil[69] and Chrysanthemum morifolium[70]
Small amounts of endogenous ethylene are also produced in mammals, including humans, due to lipid peroxidation. Some of endogenous ethylene is then oxidized to ethylene oxide, which is able to alkylate DNA and proteins, including hemoglobin (forming a specific adduct with its N-terminal valine, N-hydroxyethyl-valine).[71] Endogenous ethylene oxide, just as like environmental (exogenous) one, can alkylate guanine in DNA, forming an adduct 7-(2-hydroxyethyl)-guanine, and this poses an intrinsic carcinogenic risk.[72] It is also mutagenic.[73][74]
References
- ^ Prabhakar, Nanduri R.; Semenza, Gregg L. (September 2015). "Oxygen Sensing and Homeostasis". Physiology. 30 (5): 340–348. doi:10.1152/physiol.00022.2015. ISSN 1548-9213. PMC 4556828. PMID 26328879.
- ^ Anbalagan, Savani (17 January 2024). "Heme-based oxygen gasoreceptors". American Journal of Physiology. Endocrinology and Metabolism. 326 (2): E178–E181. doi:10.1152/ajpendo.00004.2024. PMID 38231000. S2CID 267032865.
- ^ Xu, P; Wen, X; Leal, WS (February 2020). "CO(2) per se activates carbon dioxide receptors". Insect Biochemistry and Molecular Biology. 117: 103284. doi:10.1016/j.ibmb.2019.103284. PMC 6980743. PMID 31760135.
- ^ Hogg N, Singh RJ, Kalyanaraman B (March 18, 1996). "The role of glutathione in the transport and catabolism of nitric oxide". FEBS Letters. 382 (3): 223–228. doi:10.1016/0014-5793(96)00086-5. PMID 8605974.
- ^ DeMaster EG, Quast BJ, Redfern B, Nagasawa HT (Sep 12, 1995). "Reaction of nitric oxide with the free sulfhydryl group of human serum albumin yields a sulfenic acid and nitrous oxide". Biochemistry. 34 (36): 11494–11499. doi:10.1021/bi00036a023. PMID 7547878.
- ^ Hyun J, Chaudhuri G, Fukuto JM (September 1, 1999). "The Reductive Metabolism of Nitric Oxide in Hepatocytes: Possible Interaction with Thiols". Drug Metabolism and Disposition. 27 (9): 1005–1009. PMID 10460799.
- ^ Torres, M.J.; Simon, J.; Rowley, G.; Bedmar, E.J.; Richardson, D.J.; Gates, A.J.; Delgado, M.J. (2016), "Nitrous Oxide Metabolism in Nitrate-Reducing Bacteria", Advances in Microbial Physiology, 68, Elsevier: 353–432, doi:10.1016/bs.ampbs.2016.02.007, ISBN 978-0-12-804823-8, PMID 27134026, retrieved 2021-10-14
- ^ Gillman MA, Lichtigfeld FJ (January 1981). "A comparison of the effects of morphine sulphate and nitrous oxide analgesia on chronic pain states in man". Journal of the Neurological Sciences. 49 (1): 41–45. doi:10.1016/0022-510X(81)90186-6. PMID 7205318. S2CID 32640794.
- ^ Gillman MA, Lightigfeld FJ (February 1981). "The similarity of the action of nitrous oxide and morphine". Pain. 10 (1): 110. doi:10.1016/0304-3959(81)90054-3. PMID 7232008. S2CID 53157904.
- ^ Gillman MA, Lichtigfeld FJ (May 1983). "Nitrous oxide interacts with opioid receptors: more evidence". Anesthesiology. 58 (5): 483–484. doi:10.1097/00000542-198305000-00021. PMID 6301312.
- ^ Daras C, Cantrill RC, Gillman MA (April 1983). "[3H]naloxone displacement: evidence for nitrous oxide as opioid receptor agonist". European Journal of Pharmacology. 89 (1–2): 177–178. doi:10.1016/0014-2999(83)90626-x. PMID 6305684.
- ^ Ori C, Ford-Rice F, London ED (March 1989). "Effects of nitrous oxide and halothane on mu and kappa opioid receptors in guinea-pig brain". Anesthesiology. 70 (3): 541–544. doi:10.1097/00000542-198903000-00027. PMID 2564264.
- ^ Daras, C; Cantrill, R; Gillman, MA (1983). "(3H)Naloxone displacement: evidence for nitrous oxide as opioid receptor agonist". Eur J Pharmacol. 89 (1–2): 177–178. doi:10.1016/0014-2999(83)90626-x. PMID 6305684.
- ^ Ori, C.; Ford-Rice, F; London, E.D. (1989). "Effects of nitrous oxide and halothane on mu and kappa opioid receptors in guinea-pig brain". Anesthesiology. 70 (3): 541–544. doi:10.1097/00000542-198903000-00027. PMID 2564264.
- ^ Finck, A. D., Samaniego, E., Ngai, S.H. [1995]. Nitrous oxide selectively releases met5-enkephalin and met5-enkephalin-arg6-phe7 into canine third ventricular cerebrospinal fluid. Anesthesia and Analgesia 80: 664-70
- ^ Jevtović-Todorović V, Todorović SM, Mennerick S, Powell S, Dikranian K, Benshoff N, Zorumski CF, Olney JW (Apr 1998). "Nitrous oxide (laughing gas) is an NMDA antagonist, neuroprotectant and neurotoxin". Nat Med. 4 (4): 460–463. doi:10.1038/nm0498-460. PMID 9546794. S2CID 9998244.
- ^ Christensen B, Refsum H, Garras A, Ueland PM (Jun 1992). "Homocysteine remethylation during nitrous oxide exposure of cells cultured in media containing various concentrations of folates". J Pharmacol Exp Ther. 261 (3): 1096–1105. PMID 1602376.
- ^ Koblin DD, Waskell L, Watson JE, Stokstad EL, Eger EI 2nd (Feb 1982). "Nitrous oxide inactivates methionine synthetase in human liver". Anesth Analg. 61 (2): 75–78. doi:10.1213/00000539-198202000-00001. PMID 7198880. S2CID 31136411.
- ^ Sampath V, Zhao XJ, Caughey WS (Apr 27, 2001). "Anesthetic-like interactions of nitric oxide with albumin and hemeproteins. A mechanism for control of protein function". The Journal of Biological Chemistry. 276 (17): 13635–13643. doi:10.1074/jbc.M006588200. PMID 11278308.
- ^ Dong A, Huang P, Zhao XJ, Sampath V, Caughey WS (September 30, 1994). "Characterization of sites occupied by the anesthetic nitrous oxide within proteins by infrared spectroscopy". The Journal of Biological Chemistry. 269 (39): 23911–23917. doi:10.1016/S0021-9258(19)51025-0. PMID 7929038.
- ^ Einarsdóttir O, Caughey WS (5 Jul 1988). "Interactions of the anesthetic nitrous oxide with bovine heart cytochrome c oxidase. Effects on protein structure, oxidase activity, and other properties". The Journal of Biological Chemistry. 263 (19): 9199–9205. doi:10.1016/S0021-9258(19)76525-9. PMID 2837481.
- ^ Gillman MA, Lichtigfeld FJ (March 1985). "Nitrous oxide acts directly at the mu opioid receptor". Anesthesiology. 62 (3): 375–376. doi:10.1097/00000542-198503000-00040. PMID 2983587.
- ^ Gillman MA, Lichtigfeld FJ (January 1981). "A comparison of the effects of morphine sulphate and nitrous oxide analgesia on chronic pain states in man". J. Neurol. Sci. 49 (1): 41–45. doi:10.1016/0022-510X(81)90186-6. PMID 7205318. S2CID 32640794.
- ^ Johnson, Carolyn Y. (October 16, 2009). "Poison gas may carry a medical benefit". The Boston Globe. Retrieved October 16, 2009.
- ^ Kerek F (Sep 2000). "The structure of the digitalislike and natriuretic factors identified as macrocyclic derivatives of the inorganic carbon suboxide". Hypertension Research. 23 (Suppl S33): S33–38. doi:10.1291/hypres.23.Supplement_S33. PMID 11016817.
- ^ Stimac R, Kerek F, Apell HJ (Apr 2003). "Macrocyclic carbon suboxide oligomers as potent inhibitors of the Na,K-ATPase". Annals of the New York Academy of Sciences. 986 (1): 327–329. Bibcode:2003NYASA.986..327S. doi:10.1111/j.1749-6632.2003.tb07204.x. PMID 12763840.
- ^ Kerek F, Stimac R, Apell HJ, Freudenmann F, Moroder L (23 December 2002). "Characterization of the macrocyclic carbon suboxide factors as potent Na,K-ATPase and SR Ca-ATPase inhibitors". Biochimica et Biophysica Acta (BBA) - Biomembranes. 1567 (1–2): 213–220. doi:10.1016/S0005-2736(02)00609-0. PMID 12488055.
- ^ Tubaro E. (Jun 1966). "Carbon suboxide, the probable precursor of an antitumor cellular sustance [sic]: retina". Boll Chim Farm (in Italian). 105 (6): 415–416. PMID 6005012.
- ^ Liu, D.; Jin, H; Tang, C; Du, J (2010). "Sulfur dioxide: a novel gaseous signal in the regulation of cardiovascular functions". Mini-Reviews in Medicinal Chemistry. 10 (11): 1039–1045. doi:10.2174/1389557511009011039. PMID 20540708. Archived from the original on 2013-04-26. Retrieved 2015-03-06.
- ^ Chen S, Zheng S, Liu Z, Tang C, Zhao B, Du J, Jin H (Feb 2015). "Endogenous sulfur dioxide protects against oleic acid-induced acute lung injury in association with inhibition of oxidative stress in rats". Lab. Invest. 95 (2): 142–156. doi:10.1038/labinvest.2014.147. PMID 25581610.
- ^ Tian H. (Nov 2014). "Advances in the study on endogenous sulfur dioxide in the cardiovascular system". Chin Med J. 127 (21): 3803–3807. doi:10.3760/cma.j.issn.0366-6999.20133031. PMID 25382339.
- ^ Yang R, Yang Y, Dong X, Wu X, Wei Y (Aug 2014). "Correlation between endogenous sulfur dioxide and homocysteine in children with pulmonary arterial hypertension associated with congenital heart disease". Zhonghua Er Ke Za Zhi (in Chinese). 52 (8): 625–629. PMID 25224243.
- ^ Liu D, Huang Y, Bu D, Liu AD, Holmberg L, Jia Y, Tang C, Du J, Jin H (May 2014). "Sulfur dioxide inhibits vascular smooth muscle cell proliferation via suppressing the Erk/MAP kinase pathway mediated by cAMP/PKA signaling". Cell Death Dis. 5 (5): e1251. doi:10.1038/cddis.2014.229. PMC 4047873. PMID 24853429.
- ^ Wang XB, Jin HF, Tang CS, Du JB (16 Nov 2011). "The biological effect of endogenous sulfur dioxide in the cardiovascular system". Eur J Pharmacol. 670 (1): 1–6. doi:10.1016/j.ejphar.2011.08.031. PMID 21925165.
- ^ Liang Y, Liu D, Ochs T, Tang C, Chen S, Zhang S, Geng B, Jin H, Du J (Jan 2011). "Endogenous sulfur dioxide protects against isoproterenol-induced myocardial injury and increases myocardial antioxidant capacity in rats". Lab. Invest. 91 (1): 12–23. doi:10.1038/labinvest.2010.156. PMID 20733562.
- ^ a b Borowitz JL, Gunasekar PG, Isom GE (12 Sep 1997). "Hydrogen cyanide generation by mu-opiate receptor activation: possible neuromodulatory role of endogenous cyanide". Brain Res. 768 (1–2): 294–300. doi:10.1016/S0006-8993(97)00659-8. PMID 9369328. S2CID 12277593.
- ^ Gunasekar PG, Prabhakaran K, Li L, Zhang L, Isom GE, Borowitz JL (May 2004). "Receptor mechanisms mediating cyanide generation in PC12 cells and rat brain". Neurosci Res. 49 (1): 13–18. doi:10.1016/j.neures.2004.01.006. PMID 15099699. S2CID 29850349.
- ^ Smith RP, Kruszyna H (Jan 1976). "Toxicology of some inorganic antihypertensive anions". Fed. Proc. 35 (1): 69–72. PMID 1245233.
- ^ "PubChem Substance Summary". Retrieved 7 July 2009.
- ^ Zschocke, Johannes; Georg Hoffman (2004). Vademecum Metabolism. Friedrichsdorf, Germany: Milupa GmbH.
- ^ Rose, Burton; Helmut Rennke (1994). Renal Pathophysiology. Baltimore: Williams & Wilkins. ISBN 978-0-683-07354-6.
- ^ Eszter Tuboly; Andrea Szabó; Dénes Garab; Gábor Bartha; Ágnes Janovszky; Gábor Ero″s; Anna Szabó; Árpád Mohácsi; Gábor Szabó; József Kaszaki; Miklós Ghyczy; Mihály Boros (15 January 2013). "Methane biogenesis during sodium azide-induced chemical hypoxia in rats". American Journal of Physiology. Cell Physiology. 304 (2): 207–214. doi:10.1152/ajpcell.00300.2012. PMID 23174561.
- ^ Tuboly E, Szabó A, Erős G, Mohácsi A, Szabó G, Tengölics R, Rákhely G, Boros M (Dec 2013). "Determination of endogenous methane formation by photoacoustic spectroscopy" (PDF). Journal of Breath Research. 7 (4): 046004. Bibcode:2013JBR.....7d6004T. doi:10.1088/1752-7155/7/4/046004. PMID 24185326. S2CID 206107686.
- ^ Sahakian AB, Jee SR, Pimentel M (Aug 2010). "Methane and the gastrointestinal tract". Dig Dis Sci. 55 (8): 2135–2143. doi:10.1007/s10620-009-1012-0. PMID 19830557. S2CID 5493946.
- ^ a b c Lin, Z.; Zhong, S.; Grierson, D. (2009). "Recent advances in ethylene research". J. Exp. Bot. 60 (12): 3311–36. doi:10.1093/jxb/erp204. PMID 19567479.
- ^ External Link to More on Ethylene Gassing and Carbon Dioxide Control Archived 2010-09-14 at the Wayback Machine. ne-postharvest.com
- ^ Neljubov D. (1901). "Uber die horizontale Nutation der Stengel von Pisum sativum und einiger anderen Pflanzen". Beih Bot Zentralbl. 10: 128–139.
- ^ Doubt, Sarah L. (1917). "The Response of Plants to Illuminating Gas". Botanical Gazette. 63 (3): 209–224. doi:10.1086/332006. hdl:2027/mdp.39015068299380. JSTOR 2469142. S2CID 86383905.
- ^ Gane R. (1934). "Production of ethylene by some fruits". Nature. 134 (3400): 1008. Bibcode:1934Natur.134.1008G. doi:10.1038/1341008a0. S2CID 4090009.
- ^ Crocker W, Hitchcock AE, Zimmerman PW. (1935) "Similarities in the effects of ethlyene and the plant auxins". Contrib. Boyce Thompson Inst. 7. 231-48. Auxins Cytokinins IAA Growth substances, Ethylene
- ^ Yang, S. F.; Hoffman N. E. (1984). "Ethylene biosynthesis and its regulation in higher plants". Annu. Rev. Plant Physiol. 35: 155–89. doi:10.1146/annurev.pp.35.060184.001103.
- ^ Bleecker, A. B.; Esch, J. J.; Hall, A. E.; Rodríguez, F. I.; Binder, B. M. (1998). "The ethylene-receptor family from Arabidopsis: Structure and function". Philosophical Transactions of the Royal Society B: Biological Sciences. 353 (1374): 1405–12. doi:10.1098/rstb.1998.0295. PMC 1692356. PMID 9800203.
- ^ Explaining Epinasty. planthormones.inf
- ^ Tanimoto, Mimi; Roberts, Keith; Dolan, Liam (December 1995). "Ethylene is a positive regulator of root hair development in Arabidopsis thaliana". The Plant Journal. 8 (6): 943–948. doi:10.1046/j.1365-313X.1995.8060943.x. PMID 8580964.
- ^ Zhang, Yingjiao; Du, Huan; Xu, Feiyun; Ding, Yexin; Gui, Yao; Zhang, Jianhua; Xu, Weifeng (June 2020). "Root-Bacteria Associations Boost Rhizosheath Formation in Moderately Dry Soil through Ethylene Responses". Plant Physiology. 183 (2): 780–792. doi:10.1104/pp.19.01020. PMC 7271771. PMID 32220965.
- ^ Hartman, Sjon (3 June 2020). "Trapped in the Rhizosheath: Root-Bacterial Interactions Modulate Ethylene Signaling". Plant Physiology. 183 (2): 443–444. doi:10.1104/pp.20.00379. PMC 7271798. PMID 32493810.
- ^ Dawood, Thikra; Yang, Xinping; Visser, Eric J.W.; te Beek, Tim A.H.; Kensche, Philip R.; Cristescu, Simona M.; Lee, Sangseok; Floková, Kristýna; Nguyen, Duy; Mariani, Celestina; Rieu, Ivo (April 2016). "A Co-Opted Hormonal Cascade Activates Dormant Adventitious Root Primordia upon Flooding in dulcamara". Plant Physiology. 170 (4): 2351–2364. doi:10.1104/pp.15.00773. PMC 4825138. PMID 26850278.
- ^ Negi, Sangeeta; Sukumar, Poornima; Liu, Xing; Cohen, Jerry D.; Muday, Gloria K. (January 2010). "Genetic dissection of the role of ethylene in regulating auxin-dependent lateral and adventitious root formation in tomato". The Plant Journal. 61 (1): 3–15. doi:10.1111/j.1365-313X.2009.04027.x. hdl:10339/30054. PMID 19793078.
- ^ Hartman, S; Liu, Z; van Veen, H; Vicente, J; Reinen, E; Martopawiro, S; Zhang, H; van Dongen, N; Bosman, F; Bassel, GW; Visser, EJW; Bailey-Serres, J; Theodoulou, FL; Hebelstrup, KH; Gibbs, DJ; Holdsworth, MJ; Sasidharan, R; Voesenek, LACJ (5 September 2019). "Ethylene-mediated nitric oxide depletion pre-adapts plants to hypoxia stress". Nature Communications. 10 (1): 4020. Bibcode:2019NatCo..10.4020H. doi:10.1038/s41467-019-12045-4. PMC 6728379. PMID 31488841.
- ^ van Veen et al., [1], Plant Cell, 2013
- ^ Hartman, Sjon; Sasidharan, Rashmi; Voesenek, Laurentius A. C. J. (18 January 2020). "The role of ethylene in metabolic acclimations to low oxygen". New Phytologist. 229 (1): 64–70. doi:10.1111/nph.16378. PMC 7754284. PMID 31856295.
- ^ a b Liu, Zeguang; Hartman, Sjon; van Veen, Hans; Zhang, Hongtao; Leeggangers, Hendrika A C F; Martopawiro, Shanice; Bosman, Femke; de Deugd, Florian; Su, Peng; Hummel, Maureen; Rankenberg, Tom; Hassall, Kirsty L; Bailey-Serres, Julia; Theodoulou, Frederica L; Voesenek, Laurentius A C J; Sasidharan, Rashmi (30 May 2022). "Ethylene augments root hypoxia tolerance via growth cessation and reactive oxygen species amelioration". Plant Physiology. 190 (2): 1365–1383. doi:10.1093/plphys/kiac245. PMC 9516759. PMID 35640551.
- ^ Barry, Cornelius S.; Giovannoni, James J. (6 June 2007). "Ethylene and Fruit Ripening". Journal of Plant Growth Regulation. 26 (2): 143–159. doi:10.1007/s00344-007-9002-y. S2CID 29519988.
- ^ Pandey, Bipin K.; Huang, Guoqiang; Bhosale, Rahul; Hartman, Sjon; Sturrock, Craig J.; Jose, Lottie; Martin, Olivier C.; Karady, Michal; Voesenek, Laurentius A. C. J.; Ljung, Karin; Lynch, Jonathan P.; Brown, Kathleen M.; Whalley, William R.; Mooney, Sacha J.; Zhang, Dabing; Bennett, Malcolm J. (15 January 2021). "Plant roots sense soil compaction through restricted ethylene diffusion". Science. 371 (6526): 276–280. Bibcode:2021Sci...371..276P. doi:10.1126/science.abf3013. PMID 33446554. S2CID 231606782.
- ^ Huang, Guoqiang; Kilic, Azad; Karady, Michal; Zhang, Jiao; Mehra, Poonam; Song, Xiaoyun; Sturrock, Craig J.; Zhu, Wanwan; Qin, Hua; Hartman, Sjon; Schneider, Hannah M.; Bhosale, Rahul; Dodd, Ian C.; Sharp, Robert E.; Huang, Rongfeng; Mooney, Sacha J.; Liang, Wanqi; Bennett, Malcolm J.; Zhang, Dabing; Pandey, Bipin K. (26 July 2022). "Ethylene inhibits rice root elongation in compacted soil via ABA- and auxin-mediated mechanisms". Proceedings of the National Academy of Sciences. 119 (30): e2201072119. Bibcode:2022PNAS..11901072H. doi:10.1073/pnas.2201072119. PMC 9335218. PMID 35858424.
- ^ Rosado, Daniele; Ackermann, Amanda; Spassibojko, Olya; Rossi, Magdalena; Pedmale, Ullas V (4 February 2022). "WRKY transcription factors and ethylene signaling modify root growth during the shade-avoidance response". Plant Physiology. 188 (2): 1294–1311. doi:10.1093/plphys/kiab493. PMC 8825332. PMID 34718759.
- ^ Courbier, Sarah; Hartman, Sjon (4 February 2022). "WRKYs work to limit root growth in response to shade". Plant Physiology. 188 (2): 937–938. doi:10.1093/plphys/kiab525. PMC 8825341. PMID 34791438.
- ^ Wilmowicz E, Kesy J, Kopcewicz J (December 2008). "Ethylene and ABA interactions in the regulation of flower induction in Pharbitis nil". J. Plant Physiol. 165 (18): 1917–28. doi:10.1016/j.jplph.2008.04.009. PMID 18565620.
- ^ Cockshull KE, Horridge JS (1978). "2-Chloroethylphosphonic Acid and Flower Initiation by Chrysanthemum morifolium Ramat. In Short Days and in Long Days". Journal of Horticultural Science & Biotechnology. 53 (2): 85–90. doi:10.1080/00221589.1978.11514799.
- ^ Filser JG, Denk B, Törnqvist M, Kessler W, Ehrenberg L (1992). "Pharmacokinetics of ethylene in man; body burden with ethylene oxide and hydroxyethylation of hemoglobin due to endogenous and environmental ethylene". Arch. Toxicol. 66 (3): 157–163. doi:10.1007/bf01974008. PMID 1303633. S2CID 39354680.
- ^ Bolt HM, Leutbecher M, Golka K (1997). "A note on the physiological background of the ethylene oxide adduct 7-(2-hydroxyethyl)guanine in DNA from human blood". Arch. Toxicol. 71 (11): 719–721. doi:10.1007/s002040050451. PMID 9363847. S2CID 46278715.
- ^ Csanády GA, Denk B, Pütz C, Kreuzer PE, Kessler W, Baur C, Gargas ML, Filser JG (May 15, 2000). "A physiological toxicokinetic model for exogenous and endogenous ethylene and ethylene oxide in rat, mouse, and human: formation of 2-hydroxyethyl adducts with hemoglobin and DNA". Toxicol Appl Pharmacol. 165 (1): 1–26. doi:10.1006/taap.2000.8918. PMID 10814549.
- ^ Thier R, Bolt HM (Sep 2000). "Carcinogenicity and genotoxicity of ethylene oxide: new aspects and recent advances". Crit Rev Toxicol. 30 (5): 595–608. doi:10.1080/10408440008951121. PMID 11055837. S2CID 21154564.
External links
 Media related to Gaseous signaling molecules at Wikimedia Commons
Media related to Gaseous signaling molecules at Wikimedia Commons
