| |
| Names | |
|---|---|
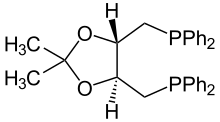
| IUPAC name
O-Isopropylidene-2,3-dihydroxy-1,4-bis(diphenylphosphino)butane
| |
| Other names
(−)-2,3-O-Isopropylidene-2,3-dihydroxy-1,4-bis(diphenylphosphino)butane
(−)-1,4-Bis(diphenylphosphino)-1,4-dideoxy-2,3-O-isopropylidene-L-threitol | |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChemSpider |
|
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C31H32O2P2 | |
| Molar mass | 498.543 g·mol−1 |
| Appearance | White solid |
| Melting point | 86 to 89 °C (187 to 192 °F; 359 to 362 K) |
| Insoluble | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
DIOP (2,3-O-isopropylidene-2,3-dihydroxy-1,4-bis(diphenylphosphino)butane) is an organophosphorus compound that is used as a chiral ligand in asymmetric catalysis. It is a white solid that is soluble in organic solvents.
DIOP is prepared from the acetonide of d,l-tartaric acid, which is reduced prior to attachment of the PPh2 substituents.
YouTube Encyclopedic
-
1/3Views:162 55336 8235 514
-
dr. cheik anta diop pt1
-
Cheikh Anta Diop: Precolonial Black Africa (Preface & Chapter 1/10)
-
Dr. John Henrik Clarke | Work of Cheikh Anta Diop
Transcription
Use
The DIOP ligand binds to metals via conformationally flexible seven-membered C4P2M chelate ring.[1][2]
DIOP is a historically important in the development of ligands for use in asymmetric catalysis, an atom-economical method for the preparation of chiral compounds. Described in 1971, it was the first example of a C2-symmetric diphosphine.[3] Its complexes have been applied to the reduction of prochiral olefins, ketones, and imines. Knowles et al. independently reported the related C2-symmetric diphosphine DIPAMP.[1]
Since the discovery of DIOP, many analogues of DIOP have been introduced. These DIOP derivatives include MOD-DIOP, Cy-DIOP, DIPAMP, and DBP-DIOP. Out of many derivatives, DBP-DIOP exhibits good regio- and enantioselectivity in the hydroformylation of butenes and styrene. DIOP was the first chiral ligand used in the platinum-tin-catalyzed hydroformylation. The reactivity, chemo – and the enantioselectivity of DIOP is influenced by CO and H2 pressure and polarity of the solvents. The best results in asymmetric hydroformylation are achieved in solvents with medium polarity: benzene and toluene.[2]
References
- ^ a b Shang, G.; Li, W.; Zhang, X. (2010). "Transition Metal-Catalyzed Homogeneous Asymmetric Hydrogenation". In Iwao Ojima (ed.). Catalytic Asymmetric Synthesis (3rd ed.). New York: John Wiley & Sons. pp. 343–436.
- ^ a b Agbossou, F.; Carpentier, J.; Mortreux, A. (1995). "Asymmetric Hydroformylation". Chem. Rev. 95 (7): 2485–2806. doi:10.1021/cr00039a008.
- ^ Dang, T. P.; Kagan, H. B. (1971). "The asymmetric synthesis of hydratropic acid and amino-acids by homogeneous catalytic hydrogenation". Journal of the Chemical Society D: Chemical Communications (10): 481. doi:10.1039/C29710000481.
