| |
| Names | |
|---|---|
| Preferred IUPAC name
Cyclooctane | |
| Other names
Cyclo-octane
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.005.484 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H16 | |
| Molar mass | 112.21 g/mol |
| Density | 0.834 g/cm3 |
| Melting point | 14.59 °C (58.26 °F; 287.74 K) |
| Boiling point | 149 °C (300 °F; 422 K) |
| 7.90 mg/L | |
| −91.4·10−6 cm3/mol | |
| Related compounds | |
Related cycloalkanes
|
Cycloheptane |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cyclooctane is a cycloalkane with the molecular formula (CH2)8.[1] It is a simple colourless hydrocarbon, but it is often a reference compound for saturated eight-membered ring compounds in general.
Cyclooctane has a camphoraceous odor.[2]
YouTube Encyclopedic
-
1/3Views:249 0506 937878
-
Naming Cycloalkanes With Substituents, Cis & Trans, Bicyclo Alkane Nomenclature
-
How to Name Cycloalkanes + 6 Examples
-
ST7● Allenes,Hemispirane,Biphenyl,Spiran,BiNepthol,Ansa,Trans cyclo octane,Helical shape
Transcription
Conformations
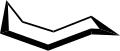
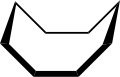
The conformation of cyclooctane has been studied extensively using computational methods. Hendrickson noted that "cyclooctane is unquestionably the conformationally most complex cycloalkane owing to the existence of many conformers of comparable energy". The boat-chair conformation (below) is the most stable form.[3] This conformation was confirmed by Allinger and co-workers.[4] The crown conformation (below)[5] is slightly less stable. Among the many compounds exhibiting the crown conformation (structure II) is S8, elemental sulfur.
Synthesis and reactions
The main route to cyclooctane derivatives involves the dimerization of butadiene, catalysed by nickel(0) complexes such as nickel bis(cyclooctadiene).[8] This process affords, among other products, 1,5-cyclooctadiene (COD), which can be hydrogenated. COD is widely used for the preparation of precatalysts for homogeneous catalysis. The activation of these catalysts under H2, produces cyclooctane, which is usually discarded or burnt:
- C8H12 + 2 H2 → C8H16
Cyclooctane participates in no reactions except those typical of other saturated hydrocarbons, combustion and free radical halogenation. Work in 2009 on alkane functionalisation, using peroxides such as dicumyl peroxide, has opened up the chemistry to some extent, allowing for example the introduction of a phenylamino group.[9]
References
- ^ Mackay, Donald (2006). Handbook of Physical-chemical Properties and Environmental Fate for Organic Chemicals. CRC Press. p. 258. ISBN 978-1-56670-687-2.
- ^ Sell, C. S. (2006). "On the Unpredictability of Odor". Angew. Chem. Int. Ed. 45 (38): 6254–6261. doi:10.1002/anie.200600782. PMID 16983730.
- ^ Hendrickson, James B. (1967). "Molecular Geometry V. Evaluation of Functions and Conformations of Medium Rings". Journal of the American Chemical Society. 89 (26): 7036–7043. doi:10.1021/ja01002a036.
- ^ Dorofeeva, O. V.; Mastryukov, V. S.; Allinger, N. L.; Almenningen, A. (1985). "The molecular structure and conformation of cyclooctane as determined by electron diffraction and molecular mechanics calculations". The Journal of Physical Chemistry. 89 (2): 252–257. doi:10.1021/j100248a015.
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "crown conformation". doi:10.1351/goldbook.C01422
- ^ a b c d e Pakes, P. W.; Rounds, T. C.; Strauss, H. L. (1981). "Conformations of cyclooctane and some related oxocanes". The Journal of Physical Chemistry. 85 (17): 2469–2475. doi:10.1021/j150617a013. ISSN 0022-3654.
- ^ Moss, G. P. (1996). "Basic terminology of stereochemistry (IUPAC Recommendations 1996)". Pure and Applied Chemistry. 68 (12): 2193–2222. doi:10.1351/pac199668122193. ISSN 0033-4545. S2CID 98272391.
- ^ Thomas Schiffer, Georg Oenbrink, “Cyclododecatriene, Cyclooctadiene, and 4-Vinylcyclohexene” in Ullmann’s Encyclopedia of Industrial Chemistry, 2005, Wiley-VCH, Weinheim. doi:10.1002/14356007.a08_205.
- ^ Deng, Guojun; Wenwen Chen; Chao-Jun Li (February 2009). "An Unusual Peroxide-Mediated Amination of Cycloalkanes with Nitroarenes". Advanced Synthesis & Catalysis. 351 (3): 353–356. doi:10.1002/adsc.200800689.