| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.017.926 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C5H13NO | |
| Molar mass | 103.16 g·mol−1 |
| Density | 0.9488 at 17 °C |
| Melting point | 38.5 °C (101.3 °F; 311.6 K) |
| Boiling point | 221.5 °C (430.7 °F; 494.6 K) |
| Hazards | |
| GHS labelling:[1] | |
 
| |
| Danger | |
| H302, H314 | |
| P260, P264, P264+P265, P270, P280, P301+P317, P301+P330+P331, P302+P361+P354, P304+P340, P305+P354+P338, P316, P317, P321, P330, P363, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
5-Amino-1-pentanol is an amino alcohol with a primary amino group and a primary hydroxy group at the ends of a linear C5-alkanes. As a derivative of the platform chemical furfural (that is easily accessible from pentoses), 5-amino-1-pentanol may become increasingly important in the future as a building block for biodegradable polyesteramides and as a starting material for valerolactam — the monomer for polyamides.
Occurrence and preparation
The complete hydrogenation of furfural (furan-2-aldehyde) yields tetrahydrofurfuryl alcohol (2-hydroxymethyltetrahydrofuran), which undergoes ring expansion upon dehydration to give dihydropyran. Dihydropyran reacts with ammonia in a reductive amination under ring opening to produce 5-amino-1-pentanol.[2]

Product yields of up to 85% can be achieved with a continuous process using a nickel-hydrotalcite catalyst.
Similarly, the hemiacetal 2-hydroxytetrahydropyran[3] that is formed from dihydropyran with hydrochloric acid can be converted to 5-amino-1-pentanol by reductive amidation with ammonia and hydrogen upon water elimination.[4]

Properties
5-Amino-1-pentanol forms white crystalline clumps at solidification temperatures around 35 °C, which dissolve in water, ethanol, and acetone.[5] The aqueous solution (500 g-l−1) reacts strongly alkaline (pH 13.2 at 20 °C).[6]
Reactions
Amino alcohols such as 5-amino-1-pentanol have been studied for their suitability of absorption of carbon dioxide.[7][8]
5-Amino-1-pentanol dehydrates when heated over ytterbium(III) oxide (Yb2O3) to give 4-penten-1-amine (I). Also formed piperidine (II), 2,3,4,5-tetrahydropyridine (III), and 1-pentylamine (IV).[9]
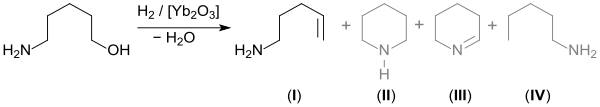
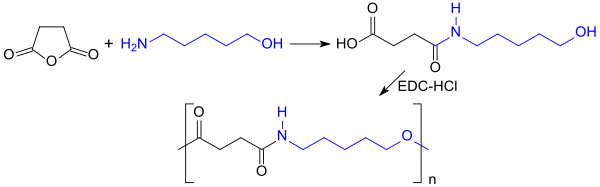
Being bifunctional, 5-amino-1-pentanol reacts in a polycondensation reaction with esters of dicarboxylic acids (or their cyclic acid anhydrides, such as succinic anhydride) to give polyesteramides. These polymers have been investigated as biodegradable plastics, e.g. absorbable sutures.[10][11] During the reaction, the succinic anhydride reacts initially with the nucleophilic amino group to form an ω-hydroxycarboxylic acid, which is subsequently polycondensed with carbodiimides, such as the hydrochloride of 1-ethyl-3-(3-dimethylaminopropyl)carbodiimides (EDC-HCl).
In a dehydrogenation, catalyzed by rhodium and ruthenium complexes, valerolactam, the δ-lactam of 5-aminopentanoic acid, is formed from 5-amino-1-pentanol in high (94%) yield.[12][13]

Valerolactam could be of relevance for polyamide 5. Polyamide 5 has garnered little attention so far but is of interest due to its ferroelectricity.[14]
References
- ^ "5-Aminopentan-1-ol". pubchem.ncbi.nlm.nih.gov.
- ^ X. Li; J. Tian; H. Liu; X. Hu; J. Zhang; C. Xia; J. Chen; H. Liu; Z. Huang (2020), "Efficient Synthesis of 5-Amino-1-pentanol from Biomass-Derived Dihydropyran over Hydrotalcite-Based Ni–Mg3AlOx Catalysts", ACS Sustain. Chem. Eng., 8 (23): 6352–6362, doi:10.1021/acssuschemeng.0c00394, S2CID 216508238
- ^ T. Oishi; M. Kanemoto; R. Swasono; N. Matsumori; M. Murata (2008), "Combinatorial Synthesis of the 1,5-Polyol System Based on Cross Metathesis: Structure Revision of Amphidinol 3", Org. Lett., 10 (22): 5203–5206, doi:10.1021/ol802168r, PMID 18959425
- ^ J. Zhang; et al. (2021), "Reductive amination of bio-based 2-hydroxytetrahydropyran to 5-amino-1-pentanol over nano-Ni-Al2O3 catalysts", New J. Chem., vol. 45, no. 9, pp. 4236–4245, doi:10.1039/D0NJ04962J, S2CID 234007765
- ^ William M. Haynes (2017), CRC Handbook of Chemistry and Physics (97th ed.), Boca Raton, FL: CRC Press, pp. 3–22, ISBN 978-1-4987-5429-3
- ^ Sigma-Aldrich Co., product no. {{{id}}}.
- ^ P. Singh; G.F. Versteeg (2008), "Structure and activity relationships for CO2 regeneration from aqueous amine-based absorbents" (PDF), Process Saf. Environ. Prot., 86 (5): 347–359, doi:10.1016/j.psep.2008.03.005
- ^ S. Oa; B.-J. Kim; J.-W. Park (2020), "Effects of carbonation on carbon dioxide capture and the mechanical properties of concrete with amine sorbents", Adv. Cement Res., 32 (11): 502–509, doi:10.1680/jadcr.18.00198, S2CID 155957571
- ^ K. Ohta; Y. Yamada; S. Sato (2016), "Dehydration of 5-amino-1-pentanol over rare earth oxides", Appl. Catal A: General, 517: 73–80, doi:10.1016/j.apcata.2016.03.001
- ^ US 4209607, S.W. Shalaby & D.D. Jamiolkowski, "Polyesteramides derived from bis-oxamidodiols and dicarboxylic acids", published 1980-6-24, assigned to Ethicon, Inc.
- ^ S.K. Murase; J. Puiggali (2014), "Poly(ester amides)s: Recent Developments on Synthesis and Applications", in S.G. Kumbar; C.T. Laurencin; M. Deng (eds.), Natural and Synthetic Biomedical Polymers, Amsterdam: Elsevier, pp. 154–166, ISBN 978-0-12-396983-5
- ^ M. Trincado; K. Kühlein; H. Grützmacher (2011), "Metal-Ligand Cooperation in the Catalytic Dehydrogenative Coupling (DHC) of Polyalcohols to Carboxylic Acid Derivatives", Chem. Eur. J., 17 (42): 11905–11913, doi:10.1002/chem.201101084, PMID 21901769
- ^ D. Pingen; D. Vogt (2014), "Amino-alcohol cyclization: Selektive synthesis of lactams and cyclic amines from amino-alcohols", Catal. Sci. Technol., 4: 47–52, doi:10.1039/C3CY00513E, S2CID 52265163
- ^ T. von Tiedemann; S. Anwas; U. Kemmer-Jonas; K. Asadi; H. Frey (2020), "Synthesis and solution processing of nylon-5 ferroelectric thin films: The renaissance of odd-nylons?", Macromol. Chem. Phys., 221 (5): 1900468, doi:10.1002/macp.201900468, hdl:21.11116/0000-0005-A118-A, S2CID 213517034
