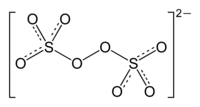
Main group peroxides are peroxide derivatives of the main group elements. Many compounds of the main group elements form peroxides, and a few are of commercial significance.[1]
YouTube Encyclopedic
-
1/3Views:25 1032 00417 662
-
Alkene + HBr + ROOR or H2O2 Peroxides Radical Reaction Mechanism - Anti Markovnikov
-
The Peroxides
-
Oxide, Peroxide, or Superoxide 006
Transcription
Examples
With thousands of tons/year being produced annually, the peroxydisulfates, S
2O2−
8, are preeminent members of this class. These salts serve as initiators for polymerization of acrylates and styrene.[1]
At one time, peroxyborates were used in detergents. These salts have been largely replaced by peroxycarbonates.
Many peroxides are not commercially valuable but are of academic interest. One example is bis(trimethylsilyl) peroxide (Me3SiOOSiMe3).[2] Phosphorus oxides form a number of peroxides, e.g. "P2O6".[3]
References
- ^ a b Jakob, Harald; Leininger, Stefan; Lehmann, Thomas; Jacobi, Sylvia; Gutewort, Sven (2007). "Peroxo Compounds, Inorganic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a19_177.pub2.
- ^ Jih Ru Hwu; Buh-Luen Chen; Santhosh F. Neelamkavil; Yuzhong Chen (2002). "Bis(trimethylsilyl) Peroxide". Encyclopedia of Reagents for Organic Synthesis. e-EROS Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/047084289X.rb219.pub3. ISBN 0-471-93623-5.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
