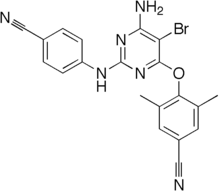
Diarylpyrimidines (DAPY) and diaryltriazines (DATA) are two closely related classes of molecules resembling the pyrimidine nucleotides found in DNA. They show great potency in inhibiting the activity of HIV reverse transcriptase.[1][2] Several compounds in this class are non-nucleoside reverse transcriptase inhibitors used clinically in the treatment of HIV/AIDS,[3][4] notably etravirine[5] and rilpivirine.[6]
YouTube Encyclopedic
-
1/2Views:3 6001 404
-
HIV-1 Reverse Transcriptase Structure and Function: Drugs by Design
-
PDB50: Celebrating PDB Contributions from Rutgers | Eddy Arnold
Transcription
References
- ^ Van Herrewege Y, Vanham G, Michiels J, Fransen K, Kestens L, Andries K, Janssen P, Lewi P (2004). "A series of diaryltriazines and diarylpyrimidines are highly potent nonnucleoside reverse transcriptase inhibitors with possible applications as microbicides". Antimicrob. Agents Chemother. 48 (10): 3684–9. doi:10.1128/AAC.48.10.3684-3689.2004. PMC 521893. PMID 15388420.
- ^ Mordant C, Schmitt B, Pasquier E, Demestre C, Queguiner L, Masungi C, Peeters A, Smeulders L, Bettens E, Hertogs K, Heeres J, Lewi P, Guillemont J (2007). "Synthesis of novel diarylpyrimidine analogues of TMC278 and their antiviral activity against HIV-1 wild-type and mutant strains". Eur J Med Chem. 42 (5): 567–79. doi:10.1016/j.ejmech.2006.11.014. PMID 17223230.
- ^ Goebel F, Yakovlev A, Pozniak AL, Vinogradova E, Boogaerts G, Hoetelmans R, de Béthune MP, Peeters M, Woodfall B (2006). "Short-term antiviral activity of TMC278--a novel NNRTI--in treatment-naive HIV-1-infected subjects". AIDS. 20 (13): 1721–6. doi:10.1097/01.aids.0000242818.65215.bd. PMID 16931936. S2CID 26078073.
- ^ Fang C, Bauman JD, Das K, Remorino A, Arnold E, Hochstrasser RM (2007). "Two-dimensional infrared spectra reveal relaxation of the nonnucleoside inhibitor TMC278 complexed with HIV-1 reverse transcriptase". Proc Natl Acad Sci USA. 105 (5): 1472–7. doi:10.1073/pnas.0709320104. PMC 2234168. PMID 18040050.
- ^ Das K, Clark AD, Lewi PJ, Heeres J, De Jonge MR, Koymans LM, Vinkers HM, Daeyaert F, Ludovici DW, Kukla MJ, De Corte B, Kavash RW, Ho CY, Ye H, Lichtenstein MA, Andries K, Pauwels R, De Béthune MP, Boyer PL, Clark P, Hughes SH, Janssen PA, Arnold E (2004). "Roles of conformational and positional adaptability in structure-based design of TMC125-R165335 (etravirine) and related non-nucleoside reverse transcriptase inhibitors that are highly potent and effective against wild-type and drug-resistant HIV-1 variants". J. Med. Chem. 47 (10): 2550–60. doi:10.1021/jm030558s. PMID 15115397.
- ^ Steve Mitchell. HIV Market To Top 10 Billion Dollars. United Press International. April 11, 2007.
