| |

| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| EC Number |
|
PubChem CID
|
|
| |
| |
| Properties | |
| C7H6FeO3 | |
| Molar mass | 193.967 g·mol−1 |
| Appearance | yellow oil |
| Melting point | 19 °C (66 °F; 292 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
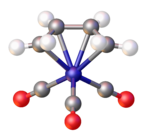
(Butadiene)iron tricarbonyl is an organoiron compound with the formula (C4H6)Fe(CO)3. It is a well-studied metal complex of butadiene.[1] An orange-colored viscous liquid that freezes just below room temperature, the compound adopts a piano stool structure.[2]
The complex was first prepared by heating iron pentacarbonyl with the diene.[3]
YouTube Encyclopedic
-
1/3Views:4534 4571 740
-
Butadiene & Cyclobutadiene complexes
-
Diene Complexes, Butadiene and Cyclobutadiene Complexes by Dr Geeta Tewari
-
Organometallic compounds: Introduction | Synthesis and structure | B.Sc | #SGKmistry Lectures
Transcription
Related compounds
Iron(0) complexes of conjugated dienes have been extensively studied. In the butadiene series, (η2-C4H6)Fe(CO)4 and (η2:η2-C4H6)(Fe(CO)4)2 have been crystallized.[4] Many related complexes are known for substituted butadienes and related species. The species (η4-isoprene)iron tricarbonyl is chiral.[5]
See also
References
- ^ Seyferth, Dietmar (2003). "(Cyclobutadiene)iron Tricarbonyl. A Case of Theory before Experiment". Organometallics. 22: 2–20. doi:10.1021/om020946c.
- ^ Reiss, Guido J. (2010). "Redetermination of (η4-s-cis-1,3-butadiene)tricarbonyliron(0)". Acta Crystallographica Section E. 66 (Pt 11): m1369. doi:10.1107/S1600536810039218. PMC 3009352. PMID 21588810.
- ^ Reihlen, Hans; Gruhl, A.; v. Heßling, G.; O. Pfrengle (1930). "Über Carbonyle und Nitrosyle. IV". Justus Liebigs Annalen der Chemie. 482: 161–182. doi:10.1002/jlac.19304820111.
- ^ Murdoch, H. D.; Weiss, E. (1962). "Butadien-Eisencarbonyl-Verbindungen (Butadieneiron Carbonyl Compounds)". Helvetica Chimica Acta. 45: 1156–61. doi:10.1002/hlca.19620450412.
- ^ Grée, R. (1989). "Acyclic Butadiene-Iron Tricarbonyl Complexes in Organic Synthesis". Synthesis. 1989 (5): 341–355. doi:10.1055/s-1989-27250. S2CID 93030781.
External links
 Media related to (Butadiene)iron tricarbonyl at Wikimedia Commons
Media related to (Butadiene)iron tricarbonyl at Wikimedia Commons
